A nuclide that lies above the band of stability shown in Figure 21.1 has too large a
Question:
A nuclide that lies above the band of stability shown in Figure 21.1 has too large a neutron-to-proton ratio to be stable. Which of the natural decay processes for such a nuclide will move it toward the band of stability?
Figure 21.1 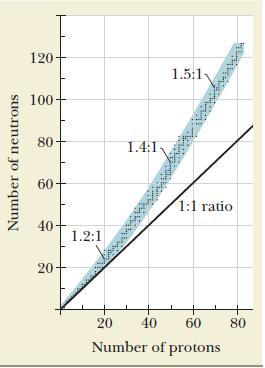
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Hassan Imtiaz
The following are details of my Professional Experience. Responsibilities Eight years of demanding teaching experience in the field of finance and business studies at Master’s Level. Completion of the given tasks within given time with quality and efficiency. Marketing professional with practical experience in and solid understanding of a diverse range of management applications, including market analysis, sales and marketing, team building and quality assurance. I have excellent skills to approach deal and sustain corporate clients / customers by demonstrating not only extraordinary communication and interpersonal skills but also high caliber presentation, negotiation and closing skills. Manage and follow up the day-to-day activities. Manage and co-ordinate the inventories. Fulfillment of all the tasks assigned.
The following are details of my Areas of Effectiveness. Finance 1. Corporate Finance 2. Advanced Corporate Finance 3. Management of Financial Institutions 4. International Financial Management 5. Investments 6. Fixed Income 7. Real Estate Investment 8. Entrepreneurial Finance 9. Derivatives 10. Alternative Investments 11. Portfolio Management 12. Financial Statement Analysis And Reporting (US GAAP & IFRS) 13. International Financial Markets 14. Public Finance 15. Personal finance 16. Real estate 17. Financial Planning Quantitative Analysis 1. Time Value Of Money 2. Statistics 3. Probability Distribution 4. Business Statistics 5. Statistical Theory and Methods Economics 1. Principles of Economics 2. Economic Theory 3. Microeconomic Principles 4. Macroeconomic Principles 5. International Monetary Economics 6. Money and Banking 7. Financial Economics 8. Population Economics 9. Behavioral Economics International Business 1. Ethics 2. Business Ethics 3. An introduction to business studies 4. Organization & Management 5. Legal Environment of Business 6. Information Systems in Organizations 7. Operations Management 8. Global Business Policies 9. Industrial Organization 10. Business Strategy 11. Information Management and Technology 12. Company Structure and Organizational Management Accounting & Auditing 1. Financial Accounting 2. Managerial Accounting 3. Accounting for strategy implementation 4. Financial accounting 5. Introduction to bookkeeping and accounting Marketing 1. Marketing Management 2. Professional Development Strategies 3. Business Communications 4. Business planning 5. Commerce & Technology Human resource management 1. General Management 2. Conflict management 3. Leadership 4. Organizational Leadership 5. Supply Chain Management 6. Law 7. Corporate Strategy Creative Writing 1. Analytical Reading & Writing Other Expertise 1. Risk Management 2. Entrepreneurship 3. Management science 4. Organizational behavior 5. Project management 6. Financial Analysis, Research & Companies Valuation 7. And any kind of Excel Queries
4.80+
150+ Reviews
230+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
A nuclide that lies below the band of stability shown in Figure 21.1 has too small a neutron-to-proton ratio to be stable. Which of the natural decay processes for such a nuclide will move it toward...
-
Tailings from uranium mining contain radium, which decays to produce radon. The radon is also radioactive and undergoes further decay. Assume the radium present is all 226 Ra, and write the pair of...
-
Both 192 Ir and 137 Cs are used in brachytherapy, the use of radioactive isotopes to treat various cancers. Both are beta emitters. (a) Explain how this observation is consistent with the information...
-
What are the uses of performance appraisal?
-
This exercise concerns the expressiveness of decision lists (Section 18.5). a. Show that decision lists can represent any Boolean function, if the size of the tests is not limited. b. Show that if...
-
Show that the Fourier series of Equation 3.89 can be expresses as Relate the coefficients cn to the an and bn of Equation 3.90. C, cos( nwt - 4,,) F(t) a, +
-
The velocity of a fluid particle moving along a horizontal streamline that coincides with the \(x\) axis in a plane, two-dimensional, incompressible flow field was experimentally found to be...
-
Consider the daily market for hot dogs in a small city. Suppose that this market is in long-run competitive equilibrium with many hot dog stands in the city, each one selling the same kind of hot...
-
3. Here are data on two companies. The T-bill rate is 4% and the market risk premium is 6%. Company $1 Discount Store Forecasted return 12% Standard deviation of returns Beta 8% 1.5 Everything $5 11%...
-
Does the neutron-to-proton ratio increase, decrease, or remain unchanged when each of the following radioactive decays occurs? (a) Beta decay (b) Positron decay (c) Gamma decay (d) Alpha decay
-
What is the binding energy per nucleon (in MeV) for 16 O, which has a mass defect of 0.1369 u?
-
Repeat Problem 9.5-15 for the anti-symmetric loading shown in the figure. Statics so reactions at A and B are equal and opposite but neither is equal to P (unlike symmetnc load case) Problem 9.5-15...
-
Your firm has 12 million shares outstanding, and you are about to issue 4 million new shares in an IPO. The IPO price has been set at $16 per share, and the underwriting spread is 8%. The IPO is a...
-
What is the break-even point for the project? How is it calculated?
-
PMF, Inc. is equally likely to have EBIT this coming year of $7 million, $13 million, or $19 million. Its corporate tax rate is 35%, and investors pay a 15% tax rate on income from equity and a 40%...
-
Describe the three techniques for feasibility analysis.
-
Craxton Engineering will either purchase or lease a new $752,000 fabricator. If purchased, the fabricator will be depreciated on a straight-line basis over seven years. Craxton can lease the...
-
Teresa owns a condominium in Florida. During the current year, she incurs the following expenses related to the property: Mortgage interest ..... $10,200 Property taxes ....... 1,500 Utilities...
-
Write each fraction as a percent. 7 50
-
In the circuit of Fig. 16.74 , find i(t) for t > 0. 4 t = 0V 6 25 F 120 V +1
-
For the circuit in Fig. 16.73 , find v(t) for t > 0. Assume that i(0) = 2 A. i(t) 10 HE v(t) 2 H 2i(t)
-
Find i 0 (t) for t > 0 in the circuit in Fig. 16.72 . 2 + Vo 1 7.5e-2t u(t) V ( +) 4.5[1 u(t)]V 0.5v. 1H
-
Find a formula for the inverse of the following function, if possible. 5 W(x) 2x-2
-
2. Apply Algorithm 9.3.8 to find a path from d to c in the road graph in Example 9.1.10 using the edge list in that example. Assume that the elements of the depth sets are put into ascending order....
-
Evaluate the impact of Capitecs leadership on the success of the strategy with regards to strategy development

Study smarter with the SolutionInn App


