A nuclide that lies below the band of stability shown in Figure 21.1 has too small a
Question:
A nuclide that lies below the band of stability shown in Figure 21.1 has too small a neutron-to-proton ratio to be stable. Which of the natural decay processes for such a nuclide will move it toward the band of stability?
Figure 21.1 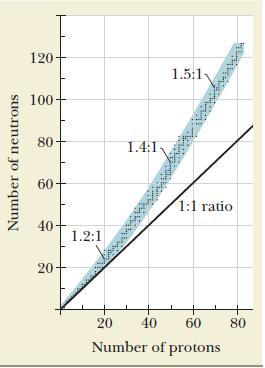
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Electr...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
A nuclide that lies above the band of stability shown in Figure 21.1 has too large a neutron-to-proton ratio to be stable. Which of the natural decay processes for such a nuclide will move it toward...
-
Tailings from uranium mining contain radium, which decays to produce radon. The radon is also radioactive and undergoes further decay. Assume the radium present is all 226 Ra, and write the pair of...
-
Radioactive decay always moves the nuclide toward the band of stability. Based on the mode of decay for each of the following radioactive nuclides, state whether it lies above or below the band of...
-
Two companies, Energen and Hastings Corporation, began operations with identical balance sheets. A year later, both required additional manufacturing capacity at a cost of $50,000. Energen obtained a...
-
For each of the following determinations write down the logical representation and explain why the determination is true (if it is): a. Zip code determines the state (U.S.). b. Design and...
-
To better understand under damped motion, use a computer to plot x(t) of Equation 3.40 (with A = 1m) and its two components [e t and cos (w1t )] and comparisons (with = 0) on the same plot as in...
-
Oil (SAE 30) at \(15.6{ }^{\circ} \mathrm{C}\) flows steadily between fixed, horizontal, parallel plates. The pressure drop per unit length along the channel is \(30 \mathrm{kPa} / \mathrm{m}\), and...
-
Bohlander Airlines is considering two alternatives for the financing of a purchase of a fleet of airplanes. These two alternatives are: 1. Issue 60,000 shares of common stock at $45 per share. (Cash...
-
Consider an investor who purchases a Treasury inflation-indexed note with an original principal amount of $100,000, a 6.500 percent annual coupon rate (coupon is paid semiannually), and 10 years to...
-
Why are the high-energy photons that accompany an electron capture decay called x rays rather than gamma rays?
-
Does the neutron-to-proton ratio increase, decrease, or remain unchanged when each of the following radioactive decays occurs? (a) Beta decay (b) Positron decay (c) Gamma decay (d) Alpha decay
-
In 1973, the GDP deflator was 15.6 in the United Kingdom and 34.3 in the United States (with 1995 = 100). In 2001, it was 116.1 in the United Kingdom and 112.1 in the United States. The exchange rate...
-
General Electric has just issued a callable 10-year, 5% coupon bond with annual coupon payments. The bond can be called at par in one year or anytime thereafter on a coupon payment date. It has a...
-
Health care is a big industry in the United States, and with the baby boomers born in the late 1940s and 1950s (after World War II) starting to retire, there will be huge demands for senior health...
-
On January 15, 2020, the U.S. Treasury issues a ten-year inflation-indexed note with a coupon of 5%. On the date of issue, the CPI is 414. By January 15, 2030, the CPI index increases to 282. What...
-
Hawar International is a shipping firm with a current share price of $6 and 15 million shares outstanding. Suppose Hawar announces plans to lower its corporate taxes by borrowing $10 million and...
-
On January 20, Metropolitan, Inc., sold 8 million shares of stock in an SEO. The current market price of Metropolitan at the time was $42.50 per share. Of the 8 million shares sold, 5 million shares...
-
Malloy Industries manufactures air conditioners. The machines used to manufacture the air conditioners usually are insulated with asbestos. Because of health risks associated with asbestos, the...
-
In Exercises 105108, evaluate each expression without using a calculator. log(ln e)
-
Using Fig. 16.85 , design a problem to help other students better understand solving for node voltages by working in the s-domain. Find the node voltages v 1 and v 2 in the circuit of Fig. 16.85...
-
Find the voltage v o (t) in the circuit of Fig. 16.84 by means of the Laplace transform. 1H ell 0.5 F 3.5u(t) A Vo
-
Find the response v(t) for t > 0 in the circuit in Fig. 16.83 . Let R = 8 Ω, L = 2 H, and C = 125 mF. v(t) 10u(t) A ell
-
a. Construct DFA of the language L = (the set of all strings with three consecutive 1's), = (0,1) (1's not necessary at the end or beginning). b. Construct an NFA without Atransitions and with a...
-
Question 1 i. ii. In the evaluation of financial statements, different end-users of these statements seek to answer different questions. Identify two questions each of the following stakeholder seek...
-
| Use the following to answer questions 13 - 17 The company's management is considering either a 100% stock dividend or a 2-for-1 stock split. Complete the following chart to assist in answering the...

Study smarter with the SolutionInn App


