Determine whether the condensation of nitromethane is spontaneous at 40 C and 1 atm. Use the thermodynamic
Question:
Determine whether the condensation of nitromethane is spontaneous at 40 °C and 1 atm. Use the thermodynamic data in Appendix G. State any assumptions you make.![]()
Thermodynamic Data From Appendix G:
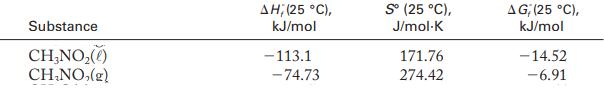
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine whether the condensation of nitromethane is spontaneous at 40 C and 1 atm we can use th...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Determine whether the vaporization of methanol is spontaneous at 80 C and 1 atm. Use the thermodynamic data in Appendix G. State any assumptions you make.
-
You are assigned to the December 31, 2011, audit of Sea Gull Airframes, Inc. The company designs and manufactures aircraft superstructures and airframe components. You observed the physical inventory...
-
For this assignment you will implement a number of Boolean functions, such as implies, nand, etc. In this assignment you will implement several Boolean functions that will later enable you to compute...
-
FIGURE Q38.5 is the current-versus-potential-difference graph for a photoelectric-effect experiment with an unknown metal. If classical physics provided the correct description of the photoelectric...
-
Assuming the earth's surface is black, estimate its temperature if the sun has an equivalent blackbody temperature of 5800 K. The diameters of the sun and earth are 1.39 x 109 and 1.29 x 107m,...
-
Calculate the expectation values of p and p2 for a particle in the state 11= 2 in a square-well potential.
-
What will happen to the vibration level of a rotating shaft if there is a mass change at one radial location?
-
Quinn Corporation is subject to tax in States G, H, and I. Quinns compensation expense includes the following. Officers' salaries are included in the payroll factor for G and I, but not for H....
-
Consider a derivative (1) of salvianolic acid B, an antioxidant and free radical scavenging compound from the root of Salvia miltiorrhiza Bunge, shown below. Answer parts i.- vii. on that molecule or...
-
At 298 K, G = -70.52 kJ for the reaction (a) Calculate G at the same temperature when P NO = 1.0 10 -4 atm, P O 2 = 2.0 10 -3 atm, and PN O2 = 0.30 atm. (b) Under the conditions in part a, in...
-
Decide whether each of the following statements is true or false. If false, rewrite it to make it true. (a) The entropy of a substance increases on going from the liquid to the vapor state at any...
-
Show that Eq. (35.14) gives zero-intensity directions that agree with Eq. (35.5).
-
Define and explain the difference between these terms: publics, shareholders, and target audiences.
-
Why is it particularly important for many out-of-home vehicles to be memorable?
-
What is the difference between IMC, advertising, and integrated brand communication (IBC)?
-
What is the use of television advertising ideally suited for?
-
How do scripts and storyboards work together to produce a television ad?
-
Refer to Short Exercise 2-3. Which of the transactions of Faith Varitek, MD, increased the total assets of the business? For each transaction, identify the asset that was increased. Apr 1 Varitek...
-
On October 1, 2014, the Dow Jones Industrial Average (DJIA) opened at 17,042 points. During that day it lost 237 points. On October 2 it lost 4 points. On October 3 it gained 209 points. Deter-mine...
-
Figure 8.20 shows a pump recirculating 300 gal/min of heavy machine tool lubricating oil at 104F to test the oils stability. The total length of 4-in pipe is 25.0 ft and the total length of 3-in pipe...
-
Linseed oil at 25C flows at 3.65 m/s in a standard hydraulic copper tube, 20 mm OD 1.2 mm wall. Compute the pressure difference between two points in the tube 17.5 m apart if the first point is 1.88...
-
A pipeline is needed to transport medium fuel oil at 77F. The pipeline needs to traverse 80 mi in total, and the initial proposal is to space pumping stations 2 mi apart. The line needs to carry 750...
-
You graduated from Deakin Business School with a major in accounting and financial planning. On graduation you have been lucky to secure a job in an accounting practice that also offers financial...
-
Hallie is appraising her subordinate, Larry's, performance. Even though Larry is a nice guy, Hallie notes that Larrys performance at work is less satisfactory than that of the other employees in the...
-
The following trial balance was extracted from the books of Night shade Ltd. as at 30 September 2020: $ Land and buildings 151,000 Motor vehicles at cost 60,000 Accumulated depreciation on motor...

Study smarter with the SolutionInn App


