Explain why the lattice energy of Na 2 O is considerably greater than that of NaF. Strategy
Question:
Explain why the lattice energy of Na2O is considerably greater than that of NaF.
Strategy
Lattice energies are evaluated by using the relationship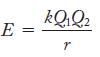
In general, the changes that can take place with the Q terms are more important than small changes in r.
Transcribed Image Text:
E = kQ1 Q₂ r
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The lattice energy of Na 2 O is greater because the greater charge on ...View the full answer

Answered By

Muhammad Ahtsham Shabbir
I am a professional freelance writer with more than 7 years’ experience in academic writing. I have a Bachelor`s Degree in Commerce and Master's Degree in Computer Science. I can provide my services in various subjects.
I have professional excellent skills in Microsoft ® Office packages such as Microsoft ® Word, Microsoft ® Excel, and Microsoft ® PowerPoint. Moreover, I have excellent research skills and outstanding analytical and critical thinking skills; a combination that I apply in every paper I handle.
I am conversant with the various citation styles, among them; APA, MLA, Chicago, Havard, and AMA. I also strive to deliver the best to my clients and in a timely manner.My work is always 100% original. I honestly understand the concern of plagiarism and its consequences. As such, I ensure that I check the assignment for any plagiarism before submission.
4.80+
392+ Reviews
587+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Explain why the lattice energy of Na 2 O is considerably greater than that of NaF. Strategy Lattice energies are evaluated by using the relationship In general, the changes that can take place with...
-
Explain why the lattice energy of Na 2 O is considerably greater than that of NaF. Strategy Lattice energies are evaluated by using the relationship In general, the changes that can take place with...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
What is the importance of developing plans to achieve organizational goals? explain with examples
-
An aluminum rod is 8.00024 m long at 20.00C. If the rod's temperature increases, it expands such that it lengthens by 0.0024% per degree temperature rise. Determine the rod's length at 28.00C and at...
-
Johnston, Inc., engaged in the following transactions involving treasury stock: Feb. 10 Purchased for cash 17,000 shares of treasury stock at a price of $25 per share. June 4 Reissued 6,000 shares of...
-
A shear layer between two fluids. Assume the following velocity distribution between two shear layers: \[v_{x}(y)=v_{0} \tanh (y / \delta)\] This is known as the Betchov and Criminale form. Calculate...
-
The Food Marketing Institute shows that 17% of households spend more than $100 per week on groceries. Assume the population proportion is p = .17 and a sample of 800 households will be selected from...
-
Longstreet Inc. has fixed operating costs of $690,000, variable costs of $3.10 per unit produced, and its product sells for $4.30 per unit. What is the company's break-even point, i.e., at what unit...
-
Use Lewis electron-dot symbols to show the electron transfer during the formation of each compound from the appropriate atoms. (a) Beryllium oxide (b) Yttrium chloride
-
Write the Lewis structure of dimethyl ether, CH 3 OCH 3 .
-
FIGURE CP15.81 shows a 200 g uniform rod pivoted at one end. The other end is attached to a horizontal spring. The spring is neither stretched nor compressed when the rod hangs straight down. What is...
-
GMS ) m IG G G WR | | | G | G github.com/Santa Barbara City College/cs105-kinneavy-f19-a04-employee-chidinmaad README.md This is where your test code will go. Replace the code in that file with...
-
Design and implement a chat server that can handle multiple clients simultaneously. Clients should be able to send messages to the server and receive updates when other clients send messages.
-
Computer Science 282 Programming Assignment #3 As always, add whatever is necessary so that the methods below behave correctly. Leave all variables, method names, and code fragments exactly as they...
-
After graduating from Hofstra University 2 years ago, you and three friends decided to start Upland Restaurant. After searching for several months for a location downtown, you decided to go a...
-
Air enters an adiabatic nozzle at 60 ft/sec, 180 psia and 480 F and, after expansion, leaves the nozzle at 15 psia. Use constant specific heats to calculate the nozzle exit velocity (use a reference...
-
True or False: 1. Only a weak statistical correlation exists between lifetime earnings and years of schooling. 2. Discovering that wages are lower for blacks and females does not prove wage...
-
What are multinational corporations (MNCs) and what economic roles do they play?
-
Compound X is a d-aldohexose that can adopt a -pyranose form with only one axial substituent. Compound X undergoes a Wohl degradation to produce an aldopentose, which is converted into an optically...
-
When d-glucose is treated with aqueous sodium hydroxide, a complex mixture of carbohydrates is formed, including d-mannose and d-fructose. Over time, almost all aldohexoses will be present in the...
-
Explain why glucose is the most common monosaccharide observed in nature.
-
A small coffee shop has opened nearby. They are looking for assistance in setting up wireless internet access for their customers. The customers should not be able to access network resources yet be...
-
Name 3 examples of network information that DHCP Server distribute to DHCP clients?
-
A user using a notebook connects to his home network and receives an email informing him that he needs to access his bank account to update his particular. An embedded link provided an email with a...

Study smarter with the SolutionInn App


