Identify the orbitals on each of the atoms that form the bonds in propylene (shown below); then
Question:
Identify the orbitals on each of the atoms that form the bonds in propylene (shown below); then indicate whether each bond is a π or a bond.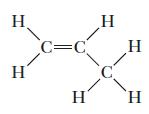
Transcribed Image Text:
H C=C H H C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The image shows the chemical structure of propylene also known as propene which is a simple alkene w...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Identify the orbitals on each of the atoms that form the bonds in H 3 CCN. How many bonds and bonds form?
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Consider the Lewis structure for glycine, the simplest amino acid: (a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each...
-
Assume that global lightning on the Earth constitutes a constant current of 1.00 kA between the ground and an atmospheric layer at potential 260 kV. (a) Find the power of terrestrial lightning. (b)...
-
Return on Equity Firm A and firm B have debttotal asset ratios of 55% and 45% and returns on total assets of 20% and 28%, respectively. Which firm has a greater return on equity?
-
Levi Strauss and Wrangler are planning new-generation jeans and must decide on the colors for their products. The possible colors are white, black, and violet. The payoff to each firm depends on the...
-
Identify the four components of a use case and how they affect each other.
-
The 12-lb slender rod AB is pinned at A and Knowing that the diameter of the cylinder is 12.5 in. and that the coefficient of static friction is 0.35 between all surfaces of contact, determine the...
-
demonstrate how four of the seven marketing mix elements the Premier Inn Hotel UK has used in the UK since 2019 reflect how it divides the market (Segmentation), who it is seeking to serve...
-
If the z axis is defined as the bond axis, draw a picture that shows the overlap of each of the following pairs of orbitals; then indicate whether a or bond forms. (a) p x , p x (b) s, p z (c) sp 2...
-
Sketch the bonds in H 2 CNH, label the type of orbital from which each bond forms, and indicate whether the bond is a or a bond.
-
When first introduced, BlackBerrys PlayBook was seen as a competitor to Apples iPad. Visit Apples website (http://www.apple.com/) and the BlackBerry website (www.blackberry.com) to learn more about...
-
After implementing the talent management model in an organization to make the hiring process more effective, what is the Post Implementation Support: (e.g. Field Support) - (What processes, tools,...
-
analyze the decision making issue for a company pursuing a new vision as a context and address the following elements: Describe the issue as it relates to that company. Use specifics to provide the...
-
Part 2 Project network/schedule -scheduling and compressing a schedule (30 points) You've been asked to manage a project that includes conducting a two-week (10 working days) computer training class....
-
Does LVMH pay much attention to cross-cultural differences in its international assignments? What should it do to better prepare its expatriates for adjustment in the host country they are sent to?
-
Which type of determinism as discussed in the screencast is most responsible for determining what happens to us in our lives? Provide at least two specific examples of this type of determinism and...
-
True or False: The discovery of a new method of birth control that is safer, cheaper, more effective, and easier to use than any other method would reduce the number of unwanted pregnancies.
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
The plastic tube is subjected to a torque of 150 N m. Determine the average shear stress in the tube if the mean dimension a = 200 mm. Each side has a thickness of t = 3 mm. 150 N-m
-
The plastic tube is subjected to a torque of 150 N m. Determine the mean dimension a of its sides if the allowable shear stress is Ï allow = 60 MPa. Each side has a thickness of t = 3 mm. 150 N-m
-
For a given maximum average shear stress, determine the factor by which the torque-carrying capacity is increased if the half-circular section is reversed from the dashed-line position to the section...
-
Summarize why fraud is easier to detect during periods of contracting money supply and harder to detect during expanding money supply. Relate your arguments to the Art Markets, Real Estate, and Oil...
-
The net changes in the statement of financial position accounts of Pharoah Inc. for the calendar year 2023 are shown below: Account Debit Credit Cash $64,300 Accounts receivable Allowance for...
-
Following is a list of internal controls. Select the COSO component that best matches each internal control. Internal control a. Implementation of policies to reduce risk of internal control failure....

Study smarter with the SolutionInn App


