In each part, an orbital diagram for an atom is given. Identify the element and whether this
Question:
In each part, an orbital diagram for an atom is given. Identify the element and whether this is the ground state of the atom. For any excited states, show the orbital diagram for the ground state.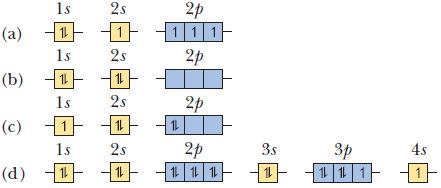
Transcribed Image Text:
1s (a) 11 1s (b) 11 1s |1| 1s 11 (c) (d) 2s 2s 11 2s 11 2s 11 2p 111 2p 12 2p 2p 12 12 12 3s 1 3p 1 1 1 4s -11-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The orbital diagrams provided depict the electron configurations of different elements in various st...View the full answer

Answered By

Shristi Singh
A freshman year metallurgy and material science student in India.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
In each part, an orbital diagram for an atom is given. Identify the element and whether this is the ground state of the atom. For any excited states, show the orbital diagram for the ground state....
-
Given the valence electron orbital level diagram and the description, identify the element or ion. a. A ground state atom b. An atom in an excited state (assume two electrons occupy the 1s orbital)...
-
Write an orbital diagram for the ground state of the phosphorus atom (Z = 15). Write all orbitals.
-
The condensed balance sheet and income statement data for Cardinal Corporation are presented below. Additional information: 1. The market price of Cardinal common stock was $5.00, $3.50, and $2.30...
-
Each of the objects shown in figure is suspended from the ceiling by a thread attached to the point marked x on the object. Describe the orientation of each suspended object with a diagram. J10 cm...
-
The trial balance of Carver, Inc., at September 30, 2010, does not balance: The accounting records hold the following errors: a. Recorded a $400 cash revenue transaction by debiting Accounts...
-
Find the condition indices and the variance decomposition proportions for the Hald cement data (Table B.21), assuming centered regressors. What can you say about multicollinearity in these data?...
-
Fisafolia Corporation has gross income from operations of $220,000 and operating expenses of $160,000 for 2014. The corporation also has $20,000 in dividends from publicly traded domestic...
-
Piedmont Company segments its business into two regions North and South. The company prepared the contribution format segmented income statement as shown: Total Company North South Sales $ 8 2 5 , 0...
-
The following transactions of Carswell Wholesale Inc. occurred in the month of September 2016: Date: Sept. 1Issued 200 common shares for $20,000. 4 To raise additional capital, Carswell borrowed...
-
Give the maximum number of electrons that may occupy the following shells or subshells. (a) The 3p subshell (b) The 4d subshell (c) The fourth principal shell (d) The third principal shell
-
The speed of sound waves in air is 344 m/s, and the frequency of middle C is 512 Hz. What is the wavelength (in m) of this sound wave?
-
The comparative balance sheets for Montalvo Company show these changes in noncash current asset accounts: accounts receivable decrease $80,000, prepaid expenses increase $28,000, and inventories...
-
In which of the following situations decision-making is easy? You have complete information You are new to a domain You are an expert in a domain Market is fluctuating
-
Which organizational structure would you prefer to work on as an employee? Least preferred? Why?
-
How can you evaluate the effectiveness of training?
-
In which model does an individual seek to make decisions with an aim to satisfy their own or an organizations interests or goals? Administrative model Political model Statistical model Classical...
-
Comment on whether external sources of recruitment such as advertisements, employment exchanges, employment agencies, educational institutes, labor contractors, recommendations, and so on will speed...
-
A telephone survey conducted by the Maritz Marketing Research company found that 43% of Americans expect to save more money next year than they saved last year. Forty-five percent of those surveyed...
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
What is the high-T approximation for rotations and vibrations? For which of these two degrees of freedom do you expect this approximation to be generally valid at room temperature?
-
In constructing the vibrational partition function, we found that the definition depended on whether zero-point energy was included in the description of the energy levels. However, the expression...
-
Although the vibrational degrees of freedom are generally not in the high-T limit, is the vibrational partition function evaluated by discrete summation?
-
Why do some firms tend to pay lower cash dividends, but also use stock repurchases in addition to distributing cash?
-
Charlene is a first-time homebuyer and has found the condo of her dreams with a purchase price of $650,000. Charlene needs to know if her savings will cover the down payment if she chooses the...
-
If the stock price of Coca Cola (KO) on the Yahoo.com website reads like this as of 4/12/2013: A) What was the closing price of the stock for the previous day? B) What is the current price of the...

Study smarter with the SolutionInn App


