In each part, arrange the orbitals in order of increasing energy in a multielectron atom. 4s (a)
Question:
In each part, arrange the orbitals in order of increasing energy in a multielectron atom.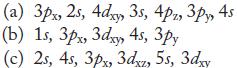
Transcribed Image Text:
4s (a) 3px, 2s, Adxy, 35, 4, 3, 45 (b) 1s, 3px, 3dxy 4s, зру (c) 2s, 4s, 3px, 3dxz, 5s, 3xy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Heres the order of increasing energy for the orbitals in each pa...View the full answer

Answered By

Shadrack Mulunga
I am a Biochemistry by profession. However, I have explored different fields of study. My quest to explore new fields has helped me gain new knowledge and skills in Business, clinical psychology, sociology, organizational behavior and general management, and Project Management. I count my expertise in Project management, in particular, creation of Work Break Down Structure (WBS) and use of Microsoft Project software as one of my greatest achievement in Freelancing industry. I have helped thousands of BSC and MSC students to complete their projects on time and cost-effectively using the MS Project tool. Generally, I find happiness in translating my knowledge and expertise to success of my clients. So far, i have helped thousands of students to not only complete their projects in time but also receive high grades in their respective courses. Quality and timely delivery are the two key aspects that define my work. All those who hired my services always come back for my service. If you hire my services today, you will surely return for more. Try me today!
5.00+
154+ Reviews
289+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
In each part, arrange the subshells in order of increasing energy in a multielectron atom. (a) 5p, 2p, 3d, 25, 3 (b) 1s, 2p, 3d, 2s, 4d, 3s (c) 1s, 2s, 3s, 2p, 3p, 4p, 3d
-
List all orbitals from 1s through 5s according to increasing energy for multielectron atoms.
-
In order to develop interesting new compounds inorganic chemists often start by noting the location of an element in the periodic table and its valence electron configuration. Suppose you are working...
-
A bank reconciliation takes time and must balance. An employee was struggling in balancing the bank reconciliation. Her supervisor told her to plug (make an unsupported entry for) the difference,...
-
Julie has been hired to help paint the trim of a building, but she is not convinced of the safety of the apparatus. A 5.0-m plank is suspended horizontally from the top of the building by ropes...
-
How might the cultural differences in the GLOBE dimensions affect how managers (a) Use work groups, (b) Develop goals/plans, (c) Reward outstanding employee performance, and (d) Deal with employee...
-
Estimate the parameters in a model for the gasoline mileage data in Table B. 3 using ridge regression. a. Use the ridge trace to select an appropriate value of \(k\). Is the resulting model adequate?...
-
Reed Corp. has set the following standard direct materials and direct labor costs per unit for the product it manufactures. Direct materials (10 lbs. @ $3 per lb.). . . . . . . . . . . . . . . . . ....
-
Let g be the function given by the formula g(x)=5x-3, defined for all the real (decimal) numbers x in the interval - 4x1. a. Make a table of values of g(x) for x= -4, -3, -2, -1, 0, and 1. b. Plot...
-
Dairy Services was organized on August 1, 20Y2. A summary of the revenue and expense transactions for August follows: Fees earned ..................................... $627,600 Wages expense...
-
The absorption spectra of ions have been used to identify the presence of the elements in the atmospheres of the Sun and other stars. (In fact, the element helium was discovered in the spectrum of...
-
For all elements with Z 10, write the electron configuration for (a) Those that have two unpaired electrons. (b) The element with the largest number of unpaired electrons. (c) Those that have only...
-
The lengths of life (in hours) of a sample of 40 6-watt light emitting diode (LED) light bulbs produced by Manufacturer A and a sample of 40 6-watt LED light bulbs produced by Manufacturer B are...
-
Let \[\begin{aligned}g_{a}^{(u)} & =\sup \left\{t: B_{t}+u t=a ight\} \\T_{a}^{(u)} & =\inf \left\{t: B_{t}+u t=a ight\}\end{aligned}\] Prove that \[\left(T_{a}^{(u)}, g_{a}^{(u)} ight)...
-
Address the questions posed in the Marks \& Spencer insert.
-
Suppose you point a pinhole camera at a \(15-\mathrm{m}\)-tall tree that is \(75 \mathrm{~m}\) away. a. If the detector is \(22 \mathrm{~cm}\) behind the pinhole, what will be the size of the tree's...
-
You have been asked to determine the power for a pump to deliver water (rwater = 62.4 lbm/ft 3 ) over a hill that is 2500 ft high at its peak to a holding tank on the other side as shown below. The...
-
In Figure Q27.12, clocks \(\mathrm{C}_{1}\) and \(C_{2}\) in frame \(S\) are synchronized. Clock \(\mathrm{C}^{\prime}\) moves at speed \(v\) relative to frame S. Clocks \(\mathrm{C}^{\prime}\) and...
-
Solve the following problems by using the binomial formula. a. If n = 4 and p = .10, find P(x = 3). b. If n = 7 and p = .80, find P(x = 4). c. If n = 10 and p = .60, find P(x 7). d. If n = 12 and p...
-
A local politician is concerned that a program for the homeless in her city is discriminating against blacks and other minorities. The following data were taken from a random sample of black and...
-
What does a partition function represent? Can you describe this term using concepts from probability theory?
-
Explain the significance of the Boltzmann distribution. What does this distribution describe?
-
Why is the probability of observing a configuration of energy different from the Boltzmann distribution vanishingly small?
-
A camera with a CCD chip of 5 x 5 mm and 1000 x 1000 sensing elements, that is equipped with a 20 mm lens, is focused on a square flat area located 0.2 m away. How many line pairs per mm will this...
-
A water park has a giant bucket that dumps water over the top of a playground (see the image below). The bucket is approximately the size of a 55-gallon drum with height 85.0 cm and diameter 57.0 cm....
-
A soccer player running on a level playing field kicks a soccer ball with a velocity of 9.4m/s at an angle of 40 above the horizontal. Determine the soccer ball's: 1. time of flight 2. range (x...

Study smarter with the SolutionInn App


