In each part, arrange the subshells in order of increasing energy in a multielectron atom. (a) 5p,
Question:
In each part, arrange the subshells in order of increasing energy in a multielectron atom.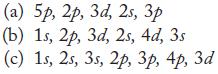
Transcribed Image Text:
(a) 5p, 2p, 3d, 25, 3 (b) 1s, 2p, 3d, 2s, 4d, 3s (c) 1s, 2s, 3s, 2p, 3p, 4p, 3d
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a 2 s 2p 3p 3d ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
In each part, arrange the orbitals in order of increasing energy in a multielectron atom. 4s (a) 3px, 2s, Adxy, 35, 4, 3, 45 (b) 1s, 3px, 3dxy 4s, (c) 2s, 4s, 3px, 3dxz, 5s, 3xy
-
Arrange each group of compounds in order of increasing acidity. (a) Phenol, ethanol, acetic acid (b) P-toluenesulfonic acid, acetic acid, chloroacetic acid (c) Benzoic acid, o-nitrobenzoic acid,...
-
Place these types of radiation in order of increasing energy per photon. (a) Green light from a mercury lamp, (b) X-rays from a dental X-ray, (c) Microwaves in a microwave oven, (d) An FM music...
-
As you might imagine, the chart of accounts for a manufacturing firm would be different from that of a service firm. Not surprisingly, service firms differ so much that software now exists for almost...
-
A cylinder of mass M and radius R rolls against a step of height h as shown in Figure. When a horizontal force F is applied to the top of the cylinder, the cylinder remains at rest. (a) What is the...
-
Describe the different ways organizations can go international.
-
Estimate the parameters in a model for the gasoline mileage data in Table B. 3 using ridge regression with the value of \(k\) determined by Eq. (9.8). Does this model differ dramatically from the one...
-
On January 1, 2010, Furball Company had Accounts Receivable $98,000 and Allowance for Doubtful Accounts $8,100. Furball Company prepares financial statements annually. During the year the following...
-
Smee Inc. produces all-natural organic peanut butter. The peanut butter is sold in 12-ounce jars. The sales budget for the first four months of the year is as follows: January February March April...
-
Propagas is used in some central heating systems where natural gas is not available. It burns according to the following equation: a. What are the chemical names for propagas and natural gas? b....
-
The absorption spectra of ions have been used to identify the presence of the elements in the atmospheres of the Sun and other stars. (In fact, the element helium was discovered in the spectrum of...
-
For all elements with Z 10, write the electron configuration for (a) Those that have two unpaired electrons. (b) The element with the largest number of unpaired electrons. (c) Those that have only...
-
The accompanying table shows data from the World Bank, World Development Indicators, for real GDP per capita (2010 U.S. dollars) in France, Japan, the United Kingdom, and the United States in 1960...
-
Let \(Z\) be a complex BM \(Z_{t}=X_{t}+i Y_{t}\). Consider the two martingales \(\left|Z_{t} ight|^{2}-2 t\) and \(\int_{0}^{t}\left(X_{s} d Y_{s}-Y_{s} d X_{s} ight)\). Prove that...
-
(1) Let \(B\) be a Brownian motion, and define the probability \(P^{b}\) via \[\left.\mathbb{P}^{b} ight|_{\mathcal{F}_{T}}:=\left.\exp \left\{-b \int_{0}^{T} B_{s} d B_{s}-\frac{b^{2}}{2}...
-
Using the balayage formula, extend the result of Proposition 4.1.7.8 when \(\varphi^{\prime}\) is replaced by a bounded Borel function. Proposition 4.1.7.8: Let \(\varphi\) be a \(C^{1}\) function....
-
As consumers increasingly use social media and mobile apps to save money, companies must go where the consumers are and offer digital coupons and other sales promotions in those locations. Digital...
-
Let \(X\) be a semi-martingale such that \(d\langle Xangle_{t}=\sigma^{2}\left(t, X_{t} ight) d t\). Assuming that the law of the r.v. \(X_{t}\) admits a density \(\varphi(t, x)\), prove that, under...
-
Solve the following problems by using the binomial tables (Table A.2). a. If n = 20 and p = .50, find P(x = 12). b. If n = 20 and p = .30, find P(x > 8). c. If n = 20 and p = .70, find P(x < 12). d....
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
How does one calculate the number of microstates associated with a given configuration?
-
Describe what is meant by the phrase the dominant configuration.
-
What is an occupation number? How is this number used to describe energy distributions?
-
The temperature dependence of the resistivity of copper and copper-nickel alloys is shown in the figure below. Explain the reason why the resistivity is increased with Ni amount. What is the residual...
-
a -B 4. [20] Two protons are separated by a distance 2a = 20 nm. At an initial time t = 0, an electron is located 10 nm above the line AB drawn between the two protons on the line CD bisecting line...
-
6) Consider an object with mass m that is thrown with an initial speed Vo at an angle with respect to the ground. The gravitational acceleration is g and it can be assumed to be constant. Derive the...

Study smarter with the SolutionInn App


