Indicate the hybridization on each central atom in the molecules with the following Lewis structures. (a) ::
Question:
Indicate the hybridization on each central atom in the molecules with the following Lewis structures.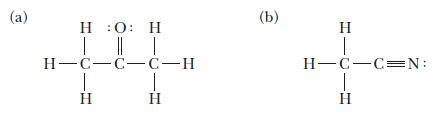
Transcribed Image Text:
(a) :: T H=C=C=C-H | (b) T H=C=C=N: H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The image youve provided shows two molecular structures labeled as a and b Lets discuss the hybridiz...View the full answer

Answered By

Charles Okinda
students should give all the instructions concerning the challenge that they face. they will get an immediate response because I am always online.
4.90+
753+ Reviews
1480+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Three resonance structures can be written for N 3 . Indicate the hybridization on the central atom for each resonance form.
-
Two resonance structures can be written for NO 2 . Indicate the hybridization on the central atom for each resonance form.
-
Give the hybridization of each central atom in the following molecules. (a) CO 2 (b) H 3 CCCH (c) H 3 CC(O)H, which has the Lewis structure H :O: H-C-C-H H
-
What type of isomers are exhibited by [Fe(en) 3 ]Cl 2 (en = ethane-1,2-diamine)? no isomers are possible. cis and trans isomers fac and mer isomers optical isomers
-
Ratios and Foreign Companies Prince Albert Canning PLC had a net loss off 18,465 on sales off 151,387 (both in thousands of pounds). What was the company's profit margin? Does the fact that these...
-
In most normal years (years in which the market has not been disrupted by Middle East wars), at each Organization of Petroleum Exporting Countries (OPEC) meeting, Saudi Arabia, the largest oil...
-
What are the basic elements of a use case diagram?
-
Tim Markus, Vice President of Phone Lines, earned $92,000 in salary last year. In addition to his salary, he also received low-interest loans from his employer. Tim's interest rate on these loans is...
-
DEF Hedge Fund has long and short positions in its portfolio. Its objective is to have zero beta exposure in its portfolio. What type of strategy does it implement? Market neutral strategy Short bias...
-
Indicate the hybridization on each central atom in the molecules with the following Lewis structures. (a) H :C: H-C-0-N-CI: 1 H (b) H T H-C=C-P-H
-
If the z axis is defi ned as the bond axis, draw a picture that shows the overlap of each of the following pairs of orbitals; then indicate whether a or bond forms. (a) p z , p z (b) p y , p y (c)...
-
Repeat Problem 39 using MATLAB. The program will do the following in one program: a. Display a root locus and pause. b. Display a close-up of the root locus where the axes go from -2 to 0.5 on the...
-
The group you are working with have come to a stand-still due to a problem that seems impossible to resolve. Members are starting to show frustration and despite much talk they are no closer to...
-
please answer this four question with APA reference for each of the question Explain the meaning of "group member assertiveness. " Why is this importantto your team? Discuss why teams assign "roles"...
-
What is the revenue recognized from gift cards during fiscal year 2020 if Best Buy collected $50 (millions) in fiscal year 2020 from new gift card sales? Consolidated Statements of Earnings $ and...
-
Describe your own philosophy of education, what education means to you and how it can be achieved, what method of learning you believe is the best, what advantages distance and personalized learning...
-
Reflect further on the group you examined in the previous reflective exercise and answer the following questions. To what extent do you consider the group an effective one in terms of the two sets of...
-
Nosmo King is an anti-smoking crusader who finds that people who don't recognize him sometimes offer him a cigarette. He always takes the cigarette and throws it away. This happens ten times a year,...
-
Saccharin is an artificial sweetener that is used in diet beverages. In order for it to be metabolized by the body, it must pass into cells. Below are shown the two forms of saccharin. Saccharin has...
-
The steel step shaft has an allowable shear stress of Ï allow = 8 MPa. If the transition between the cross sections has a radius r = 4 mm, determine the maximum torque T that can be applied. 50...
-
The symmetric tube is made from a high-strength steel, having the mean dimensions shown and a thickness of 5 mm. If it is subjected to a torque of T = 40 N m, determine the average shear stress...
-
The 304 stainless steel tube has a thickness of 10 mm. If the applied torque is T = 50 N # m, determine the average shear stress in the tube. The mean dimensions are shown. 30 mm 70 mm
-
Tim has to wear more than one kind of protective eyewear for his job. What are the requirements in this case?
-
7) The XYZ Company has estimated expected cash flows for 1996 to be as follows: Probability Cash flow .10 $120,000 .15 140,000 .50 150,000 .15 180,000 .10 210,000 Calculate: a. expected value b....
-
In what industry is Macy's Inc participating? Do they hold a large percentage of market share? In either scenario, how much market share does this firm currently hold?

Study smarter with the SolutionInn App


