Indicate the hybridization on each central atom in the molecules with the following Lewis structures. (a) H
Question:
Indicate the hybridization on each central atom in the molecules with the following Lewis structures.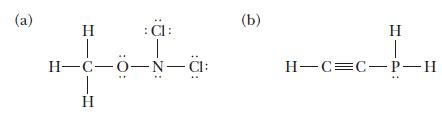
Transcribed Image Text:
(a) H :C: H-C-0-N-CI: 1 H (b) H T H-C=C-P-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a C sp ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Three resonance structures can be written for N 3 . Indicate the hybridization on the central atom for each resonance form.
-
Two resonance structures can be written for NO 2 . Indicate the hybridization on the central atom for each resonance form.
-
Give the hybridization of each central atom in the following molecules. (a) CO 2 (b) H 3 CCCH (c) H 3 CC(O)H, which has the Lewis structure H :O: H-C-C-H H
-
a) If the block is at rest (and the only forces acting on the block are the force due to gravity and the normal force from the table), what is the magnitude of the force due to friction? b) Suppose...
-
Du Pont Identity Construct the Du Pont identity for Smolira Golf Corp.
-
A computer hardware firm sells both laptop computers and printers. It has a large inventory of laptops and printers that it wants to sell, so it has no variable production cost. Through the magic of...
-
Describe normal flow, sub-flow, and alternate flow. How do they differ?
-
1. As of 2017, what type of manufacturing process did LWT appear to be using? What level of customization was it offering? Where was the point of customization? 2. Using Table 3.2 and Figure 3.12 as...
-
The following information is available for Baker Industries: Cost of goods manufactured Beginning finished goods inventory Ending finished goods inventory Required: Compute the cost of goods sold....
-
Nitrous acid has the skeleton structure HONO. What are the hybrid orbitals on the nitrogen atom and the central oxygen atom?
-
Indicate the hybridization on each central atom in the molecules with the following Lewis structures. (a) :: T H=C=C=C-H | (b) T H=C=C=N: H
-
The current price of a stock share that pays no dividend is \( 50\). The price follows a GBM with drift \(12 \%\) and volatility \(35 \%\); the continuously compounded risk-free rate is \(5 \%\)....
-
An insulated beaker with negligible mass contains liquid water with a mass of 0.205 kg and a temperature of 83.9 C Part A How much ice at a temperature of -10.0 C must be dropped into the water so...
-
We need to make marketing plan for chobani in the Philippines. (Focus on Philippines only) This is the outlines. (Each part consist of 20o words) Positioning - Philippines: Describe the intended...
-
Greene states that "the normative and descriptive Trolley Problems are closely related." What are the normative and descriptive Trolley problems, and how does Greene think they are related?
-
According the Florida Caribbean Cruise Association in 2016 the Caribbean captured 34% of the cruise market. Despite this control, the cruise industry continues to create many issues for Caribbean...
-
Complete the self-assessment online quiz: Moment of Truth: Online Ethics Quiz Links to an external site.(Media Partners, 2018)Links to an external site. Informed by the Quiz results, outline a plan...
-
The following item appeared in a major daily newspaper: Does this observation in fact violate the laws of supply anddemand? HOME PRICES HOME SALES Though sales are down, prices continue to rise in...
-
Determine the reactions in supports A and D and connections B and C. Sketch its shear and moment diagram and determine the magnitude ankoration of the maximum shear and moment for every member. 18 3...
-
The shaft is used to transmit 30 hp while turning at 600 rpm. Determine the maximum shear stress in the shaft. The segments are connected together using a fillet weld having a radius of 0.18 in. 2,5...
-
The built-up shaft is designed to rotate at 450 rpm. If the radius of the fillet weld connecting the shafts is r = 13.2 mm, and the allowable shear stress for the material is Ï allow = 150 MPa,...
-
The built-up shaft is to be designed to rotate at 450 rpm while transmitting 230 kW of power. Is this possible? The allowable shear stress is Ï allow = 150 MPa. 100 mm 60 mm
-
What are some of the main takeaways therapists would want their trauma clients to know before the session ends? explain
-
In a bilateral monopoly, what happens when the buyer and seller cannot agree on a price? Explain.
-
Raider sailboats demand is listed below. The forcast for year 5 is 5 , 6 0 0 sailboats, what will be the demand level for spring of year 5 ?

Study smarter with the SolutionInn App


