Partial information is given in each column in the following table. Fill in the blank spaces. Symbol
Question:
Partial information is given in each column in the following table. Fill in the blank spaces.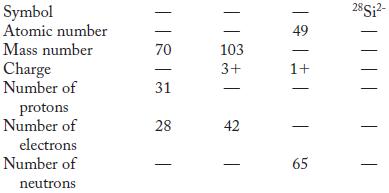
Transcribed Image Text:
Symbol Atomic number Mass number Charge Number of protons Number of electrons Number of neutrons. 70 31 28 | || 103 3+ - - - 49 1+ 65 28Si²-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Symbol Atomic number Mass number Charge N...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The following table summarizes the facts of five independent cases (labeled a through e) of U.S. companies engaging in credit transactions with foreign corporations while the foreign exchange rate is...
-
Start of Payroll Project 7-3a October 9, 20-- No. 1 The first payroll in October covered the two workweeks that ended on September 26 and October 3. This payroll transaction has been entered for you...
-
Given the partial information in each column of the following table, fill in the blanks. Symbol Atomic number Mass number Charge Number of protons Number of electrons Number of neutrons 11 10 12...
-
Four different processes for baking Oreo cookies are considered for the 2008 season. The cookies produced by each process are evaluated in terms of their overall quality. Since the cookies sometimes...
-
The 2010 accounting records of Verlander Transport reveal these transactions and events. InstructionsPrepare the cash flows from operating activities section using the direct method. (Not all of the...
-
Use a computer algebra system to find the volume of the solid bounded by the graphs of the equations. z = 9 - x 2 - y 2 z = 0
-
Mcfadzean and Honarzad run a farmers' market stall as a partnership. Honarzad withdrew \($26\) 300 cash on 25 October 2025. Mcfadzean withdrew \($17\) 500 on 28 November 2025. On 1 April 2026...
-
Chesterfield County had the following transactions. Prepare the entries first for fund-based financial statements and then for government-wide financial statements. a. A budget is passed for all...
-
56. If the maximum concentration of PbCl2 in water is 0.01M at 298 K. Its maximum concentration in 0.1M NaCl will be : (1) 4 103 M x (3) 4 102 M (2) 0.4 10M x (4) 410 M
-
Write the formula of each of the following compounds, and indicate whether each is ionic or molecular. (a) Calcium phosphate (b) Germanium dioxide (c) Iron(III) sulfate (d) Phosphorus tribromide
-
Name each of the following compounds, and indicate whether each is ionic or molecular. (a) NO (b) Y 2 (SO 4 ) 3 (c) Na 2 O (d) NBr 3
-
Use Internet sites to answer the following questions: Provide the links to three News & Topics sites provided by Tax Sites for states (www.taxsites.com).
-
Of the following mixture, which do you think has entropy departure function of larger magnitude (a) 50 mol% methane mixed with 50 mol% ethane (b) 50 mol% acetone mixed with 50 mol% chloroform?...
-
Tybo Corporation adjusts its debt so that its interest expenses are 21% of its free cash flow. Tybo is considering an expansion that will generate free cash flows of $2.16 million this year and is...
-
Explain the increased popularity of continuous improvements and work process engineering in the past twenty years.
-
Why is strategic audit recommended in case study analysis for students?
-
For an organization that you know well, what changes will it have to face up to in the near future? What is the organization's capacity for change and how would you recommend it approaches the...
-
Is the presentation of a personal income statement appropriate?
-
You have accepted the engagement of auditing the financial statements of the C. Reis Company, a small manufacturing firm that has been your auditee for several years. Because you were busy writing...
-
A compound with molecular formula C 5 H 10 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR lle 2.0 1.5 0.5 ppm 4.5 4.0 33.2 3.5 3.0 2.5 1.0...
-
A compound with molecular formula C 10 H 10 O has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR 10 3 1 ppm 4 87.1| 18.7 Integration Values 17.1...
-
A compound with molecular formula C 4 H 6 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR 25 2.0 2.0 1.5 ppm 3.0 Integration Values 5.0 4.5...
-
Use a trigonometric ratio to solve for d. Round to two decimal places as necessary. Show your work here d= d 13 12
-
Describe the impact that diversity awareness has on giving and receiving feedback when communicating and collaborating with others. Describe how you can use strategies for interacting with diverse...
-
Given the following declaration of the "textBox1" object. Which can place the "textBox1" object at the location (210, 175)? Given the following declaration of the "textBox1" object. Which can place...

Study smarter with the SolutionInn App


