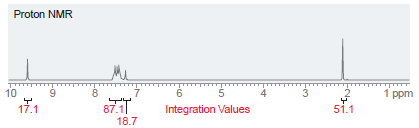
A compound with molecular formula C 10 H 10 O has the following NMR spectrum. Determine the
Question:
Transcribed Image Text:
Proton NMR 10 3 1 ppm 4 87.1| 18.7 Integration Values 17.1 51.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The signal at 96 ppm represents one ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 13 H 10 O produces a strong signal at 1660 cm 1 in its IR spectrum. The 13 C NMR spectrum for this compound is shown below. Identify the structure of this...
-
A compound with molecular formula C 11 H 14 O 2 exhibits the following spectra ( 1 H NMR, 13 C NMR, and IR). Identify the structure of this compound. Proton NMR 11 Chemical Shift (ppm) Carbon NMR...
-
Identify the following compounds. (Relative integrals are given from left to right across the spectrum.) a. The 1H NMR spectrum of a compound with molecular formula C4H10O2 has two singlets with an...
-
A teacher traces a small circle on the palm of a kindergartener's hand to let him know it is time for free play. What type of disability does this child most likely have? A. Traumatic Brain Injury B....
-
"Returns were much higher in the 1970s than in the early 1990s because the interest rates were much higher." True or false?
-
Quantum Logistics, Inc., a wholesale distributor, is considering the construction of a new warehouse to serve the southeastern geographic region near the AlabamaGeorgia border. There are three cities...
-
With reference to Exercise 3.34, suppose that the questionnaire filled in by one of the 150 persons is to be double-checked. If it is chosen in such a way that each questionnaire has a probability of...
-
Using the following accounts, prepare a classified balance sheet at year end, May 31, 2014: Accounts Payable, $1,600; Accounts Receivable, $2,200; Accumulated DepreciationEquipment, $1,400; Cash,...
-
Suppose you want to sell naked options on a bitcoin miner like MARA or RIOT. Could you hedge by also buying some bitcoin at a broker, or COINBASE? You would have to be very very explicit, showing a...
-
Assume that it is now May 13, 1994 and that the yield curve has changed accordingly (see Table 3.7). (a) What is the value of the unhedged portfolio now? (b) What is the value of the hedged...
-
A compound with molecular formula C 5 H 10 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR lle 2.0 1.5 0.5 ppm 4.5 4.0 33.2 3.5 3.0 2.5 1.0...
-
A compound with molecular formula C 4 H 6 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR 25 2.0 2.0 1.5 ppm 3.0 Integration Values 5.0 4.5...
-
Calculate the vapor pressure of water at 150C using the van der Waals equation of state. Repeat the calculation at 370C. How do these values compare with values obtained with the IF97 model?
-
As part of your PPE activities, the PPE manager asked you to help her with an upcoming coding audit she needs to perform. There is an opportunity for a vacant position at the facility, as a Coding...
-
You are CEO of SUNSUP Corporation, a major innovator and manufacturer of solar panels. As part of SUNSUP's commitment to social responsibility, you initiated and led, between 2017 and 2022, the...
-
Are there proactive actions that human resources could take to avoid such situations? What would you have done differently in managing the situation? Are there any legal implications/considerations?
-
Identify potentially applicable employment laws Identify applicable laws even if more information is needed to determine applicability with certainty. State whether the facts suggest that the law was...
-
One of your nurses was involved in a car accident. The nurse has recovered and wants to return to work. The nurse informs you that because of the accident they are not able to lift more than 20...
-
(a) Draw a scatter diagram. (b) Determine the value of r, rounded to the nearest thousandth. (c) Determine whether a correlation exists at = 0.05. (d) Determine whether a correlation exists at a =...
-
Juanita owns a home in Richardson, TX. She purchases a Homeowners Policy (HO-3) from Farm State Ins. Co. The policy provides $100,000 in liability coverage (coverage E) and $5,000 in Med Pay coverage...
-
We?ll see that there are two isomeric substances both named 1, 2-dimethylcyclohexane. Explain. -C3 1,2-Dimethylcyclohexane CH
-
Give IUPAC names for the following cycloalkanes: (c) CH2CH2CH3 (b) CH (a) CH CH3 (f) Br (e) C CH-CH (d) CHICH3)2 "CH C(CH3)3 Br
-
Draw structures corresponding to the following IUPAC names: (a) 1, 1-Dimethylcycloocatne (b) 2-Cyclobutylhexane (c) 1, 2-Dichlorocyclopentane (d) 1, 3-Dibromo-5-methylcyclohexane
-
Consider the following information: State Probability ABC Inc.'s Return Boom 0.25 15% Normal 0.50 8% Slowdown 0.15 4% Recession 0.10 -3% i. 11. iii. What is the expected return? What is the variance?...
-
The mythical country of Quitar has just established a policy to give very generous in-kind benefits to the nation's poor, currently defined as those earning less than $10,000 a year. Several years...
-
Question 1 The external loadings imposed on a horizontal beam is shown in Figure Q1(a). The beam is pivoted at Point A. (a) (b) 100 N/m 50 N/m 20 N 40 cm 50 cm Figure Q1 70 cm Apply the principle of...

Study smarter with the SolutionInn App


