State whether increasing temperature increases or decreases the value of the equilibrium constant for the following reactions.
Question:
State whether increasing temperature increases or decreases the value of the equilibrium constant for the following reactions.
For each reaction, an equilibrium constant at 298 K is given. Calculate ΔG ° for each reaction.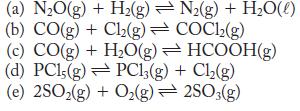
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a K eq decreases with increasing temperature ...View the full answer

Answered By

Krishnavendra Y
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagiarism), well-researched and critically analyzed papers.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
State whether increasing temperature increases or decreases the value of the equilibrium constant for the following reactions. For each reaction, an equilibrium constant at 298 K is given. Calculate...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
2. A firm's or cash flow, is its profits after deductions for all expenses, including or wear and tear on capital goods such as machinery.
-
A sphere is suspended in air in a dark room and maintained at a uniform incandescent temperature. When first viewed with the naked eye, the sphere appears to be brighter around the rim. After several...
-
The following temperature/composition data were obtained for a mixture of two liquids A and B at 1.00 atm, where x is the mole fraction in the liquid and Y the mole fraction in the vapour at...
-
Because both radial readings on the top of a vertical motor are horizontal, how can you differentiate them?
-
Higgins Company produced 50,000 units, 10,000 more than budgeted. Production data are as follows. Except for physical units, all quantities are in dollars. 1. Fill in the unknowns. 2. Give a brief...
-
Calculate the maximum stress in a 5 mm thick steel plate with an 1 8 mm diameter round hole, given h gross = 1 2 cm and the tensile load is 8 . 5 kN . ( Final answer in MPa ) P h gross d
-
Calculate the vapor pressure of each of the following at the given temperature. For each reaction, an equilibrium constant at 298 K is given. Calculate G for each reaction.
-
Calculate G and G at 37 C for the following equation. For each reaction, an equilibrium constant at 298 K is given. Calculate G for each reaction.
-
Refer to the Country Designs data set. Country Designs is a manufacturer of large flower pots for urban settings. The company has these standards: Country Designs allocated fixed manufacturing...
-
What is typically shown/seen on social media sites?
-
Internet marketing, like other vehicles we have looked at, has a diverse array of advertising options. What are some of the most common, and how can they each help to visually and verbally express...
-
What makes guerrilla marketing an active versus passive form of advertising?
-
What are the strategic uses of alternative media in an IMC campaign?
-
What is cause marketing, and why is it important to have it be a part of an IMC campaign?
-
The trial balance of Garvey, Inc., at September 30, 2012, does not balance: The accounting records hold the following errors: a. Recorded a $100 cash revenue transaction by debiting Accounts...
-
Data 9.2 on page 540 introduces the dataset Cereal, which includes information on the number of grams of fiber in a serving for 30 different breakfast cereals. The cereals come from three different...
-
Find the volume flow rate of water exiting from the tank shown in Fig. 7.12. The tank is sealed with a pressure of 140 kPa above the water. There is an energy loss of 2.0 Nm / N as the water flows...
-
Water at 40°F is flowing downward through the fabricated reducer shown in Fig. 7.11. At point A the velocity is 10 ft/s and the pressure is 60 psig. The energy loss between points A and B is 25...
-
A horizontal pipe carries oil with a specific gravity of 0.83. If two pressure gages along the pipe read 74.6 psig and 62.2 psig, respectively, calculate the energy loss between the two gages.
-
On December 31, 2021, Kona purchased debt securities as trading securities. Pertinent data are as follows:\ Fair Value\ Security Cost At 12/31/22\ A $225,000 $215,000\ B 200,000 210,000\ C 230,000...
-
A company's inventory records report the following in November of the current year: Date Activities Units Acquired at Cost Units Sold at Retail November 1 Beginning inventory 5 units @ $ 4 4 = $ 2 2...
-
Salmone Company reported the following purchases and sales of its only product. Salmone uses a perpetual inventory system. Determine the cost assigned to cost of goods sold using FIFO. Date...

Study smarter with the SolutionInn App


