State which compound in each of the following pairs you expect to have the higher boiling point.
Question:
State which compound in each of the following pairs you expect to have the higher boiling point. Explain your answer.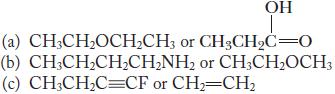
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a CH 3 CH 2 COOH hydrogen b...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
State which compound in each of the following pairs you expect to have the higher boiling point. Explain your answer.
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
Did Hopkins materially misrepresent his health when applying for insurance? Does Golden Rule have the right to rescind his insurance policies?
-
Let the sequence [x (n T)) denote die input applied to a tapped-delay-line equalizer. Show char inter-symbol interference is eliminated completely by the equalizer provided that its frequency...
-
A metal rod of length 2L, diameter D, and thermal conductivity k is inserted into a perfectly insulating wall, exposing one-half of its length to an air stream that is of temperature T and provides a...
-
Water at \(10{ }^{\circ} \mathrm{C}\) flows through a smooth \(60-\mathrm{mm}\)-diameter pipe with an average velocity of \(8 \mathrm{~m} / \mathrm{s}\). Would a layer of rust of height \(0.005...
-
The Park Plaza Hotel awarded its valet and laundry concession to Larson for a three-year term. The contract contained the following provision: It is distinctly understood and agreed that the services...
-
Gleason purchased a used van for use in its business on January 1, 2020. It paid $9,000 for the van. Gleason expects the van to have a useful life of four years, with an estimated residual value of...
-
Write the structural formula and name the organic product expected from the acid-catalyzed condensation reaction of CH 3 OH.
-
Draw all the isomers of trichlorobenzene.
-
Provide examples of two controls that improve completeness over recording of acquisitions transactions.
-
Suppose that firms are expecting 6 percent inflation while workers are expecting 9 percent inflation. How much of a pay raise will workers demand if their goal is to maintain the purchasing power of...
-
Explain the elements of the CCCER/5C model.
-
Irving owns a chain of movie theaters. He is considering whether he should build a new theater downtown. The expected rate of return is 15 percent per year. He can borrow money at a 12 percent...
-
Explain how an organization can demonstrate its adherence with the principle that management decisions are based on a long-term philosophy (principle 1).
-
Aggregate supply shocks can cause _______ inflation rates that are accompanied by _______ unemployment rates. a. higher; higher b. higher; lower c. lower; higher d. lower; lower
-
Izzy is an employee of Kosmo's Kustom Kars, Inc. The company rebuilds classic automobiles for resale. Last year, Izzy bought a rebuilt 1956 Thunderbird for $15,000 from the company. A car like Izzy's...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
An optical filter can be described by a Jones matrix Obtain the form of the emerging light for each of the following incident beams: (a) A plane polarized beam polarized at angle θ to...
-
Keeping Eq. (8.33) in mind, write an expression for the randomly polarized flux density component (I n ) of a partially polarized beam in terms of the Stokes parameters. To check your result, add a...
-
Construct a Mueller matrix for an isotropic plate of absorbing material having an amplitude transmission coefficient of t. What Mueller matrix will completely depolarize any wave without affecting...
-
DANELLA DANCE studios is evaluating the purchase of new sound equipment that will increase the probability of your aerobic dances. The equipment will cost $28,000. Danells is not sure how many...
-
In your initial post of including references in APA , describe three ways in which data are used in business functions. Use one way data is used in Accounting, one way in Marketing and one way in...
-
WHY IS CREDIT CALLED AS A PRODUCTIVE ASSET THAT MUST BE MANAGED WITH A BANK'S PROPERTIES?

Study smarter with the SolutionInn App


