State which compound in each of the following pairs you expect to have the higher boiling point.
Question:
State which compound in each of the following pairs you expect to have the higher boiling point. Explain your answer.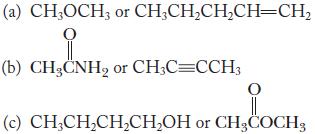
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To determine which compound in each pair will have the higher boiling point we need to consider the ...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
State which compound in each of the following pairs you expect to have the higher boiling point. Explain your answer.
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
Good Morning Food, Inc. is using the profitability index (PI) when evaluating projects. You have to find the PI for the company's project, assuming the company's cost of capital is 14.29 percent. The...
-
The step-size parameter plays a critical role in the operation of the LMS algorithm. In this context, discuss the following two issues: (a) Stability. If exceeds a certain critical value, the...
-
In one segment of the TV series Myth-Busters, an experiment was conducted to test the common belief that people are more likely to yawn when they see others yawning. In one group, 34 subjects were...
-
The John Gore Organization owns and operates the Charles Playhouse, a theater in Boston, Massachusetts. Evelyn Castillo has diabetes, a disability under the Americans with Disabilities Act (ADA)....
-
Alternative methods of joint-cost allocation, product-mix decisions. The Southern Oil Company buys crude vegetable oil. Refining this oil results in four products at the splitoff point: A, B, C, and...
-
2 Charlie's Crispy Chicken (CCC) operates a fast-food restaurant. When accounting for its first year of business, CCC created several accounts. Account Name Description Balance Accounts Payable...
-
Write the structural formula and name the organic product expected from the acid-catalyzed condensation reaction of CH 3 OH.
-
Draw all the isomers of trichlorobenzene.
-
The company began the year with a balance in its long-term debt account of $233,000. During the year, additional debt of $172,000 was incurred. The ending balance in the long-term debt account was...
-
Dolf Schulman asked for your advice on several issues facing Almond Chemical's partner company, Chongquing No. 2 Chemical Company. Agreement was reached in several areas: They will continue to place...
-
1.Imagine that you are a new hire at a commercial bank who has just completed her first performance review. Given that this entity believes in using the most modern approaches and systems, discuss...
-
During your meeting with Clarice the following day, she tells you the following: "Michael is weird and he creeps me out. He never says hello to anyone or joins in co-worker chit chat. But then, out...
-
A recent college graduate is looking to begin saving for retirement. Option A is a savings account with 3.5% annual simple interest. Option B is a savings account with 3.5% annual interest compounded...
-
Using the Moderna 2022 annual report Moderna-2022-Annual-ReportvF.pdf (q4cdn.com)Are, big data being used in the above industry? A) How is big data used in clinical trials, vaccine development,...
-
Modern Convenience specializes in making robotic conveyor systems to move materials within a factory. Model #89 accounts for approximately 60 percent of the companys annual sales. Because the company...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Will we get an interference pattern in Youngs Experiment (Fig. 9.11) if we replace the source slit S by a single long-filament lightbulb? What would occur if we replaced the slits S 1 and S 2 by...
-
A liquid cell containing an optically active sugar solution has a Jones matrix given by (a) Determine the polarization of the emerging light if the incident beam is a horizontal P-state. (b)...
-
Two linear optical filters have Jones matrices and Identify these filters. 1 eim/4 1 A2 pT/4
-
On April 1, 2022, Green Corporation issued 6% bonds dated February 1, 2022, with the face amount of $500,000. The bonds were sold for their present value on April 1, 2022, plus two months of accrued...
-
If $6,000 was paid for six months' worth of rent, what amount would you debit to rent expense at the end of every month?
-
Steve Lenda,are sident taxpayer,sold the following CGT assets during the 2O21/22tax year: ASSET COST BASE ACQUISITION DATE DISPOSAL DATE SALE PRICE Shares-A Grade $29,OOO 27Dec2O 7Oct21 $44,OOO Boat...

Study smarter with the SolutionInn App


