The compound B 10 H 14 has an unusual structure, with some of the hydrogen atoms bridging
Question:
The compound B10H14 has an unusual structure, with some of the hydrogen atoms bridging between two of the boron atoms. What is the molecular mass of B10H14?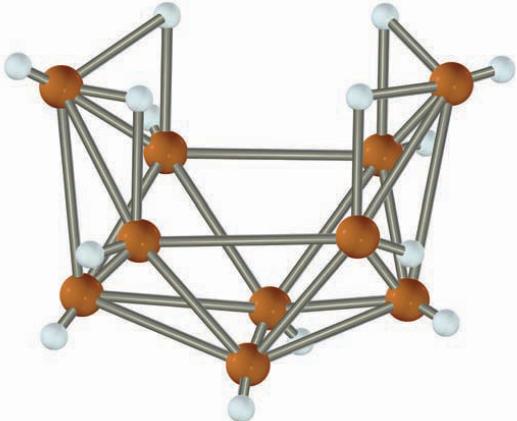
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Molar mass or molecular mass is calculated from the summation o...View the full answer

Answered By

Hillary Waliaulah
As a tutor, I am that experienced with over 5 years. With this, I am capable of handling a variety of subjects.
5.00+
17+ Reviews
30+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
small, white atoms are hydrogens, grey atoms are carbons, red atoms are oxygens what is the molecular formula for this compound and then what is its molecular mass this is a malodorous bloodred...
-
Boron is a group IIIA element. Open the molecular model for boron trifluoride from the 3D Molecular Models section of the book's website. Near the boron atom, above and below the plane of the atoms...
-
Company OMEGA wants to invest in Bonds and Stocks. The financial manager was looking at different types of Bonds and Stocks. After studying the market, he decided to choose 3 bonds and 3 stocks....
-
A spherical hole of radius a in a conducting medium can serve as an electromagnetic resonant cavity. (a) Assuming infinite conductivity, determine the transcendental equations for the characteristic...
-
A certain gas station sells X thousand gallons of gas each week. Suppose that the cumulative distribution function for X is F (x) = 1 - 1/4 (2 - x) 2 , 0 x 2. (a) If the tank contains 1.6 thousand...
-
A used car dealer sold an automobile to a customer (Robert) on an installment basis. The installment sales contract calls for Robert to make monthly payments of principal and interest. The contract...
-
Edwards has decided to use monetary- unit sampling in the audit of an entitys accounts receivable balance. Few, if any, misstatements of account balance overstatement are expected. Required: a....
-
Kericho Sports Club has been operating for some time now. The treasurer analysed the cash book and produced the following Receipts and Payments account for the year ended 31st Dec. 2018. Receipts...
-
Write the symbol for the monatomic ion that is expected for each of the following elements. (a) Iodine (b) Magnesium (c) Oxygen (d) Sodium
-
Aspartame is an artificial sweetener that has the formula C 14 H 18 N 2 O 5 . What is the molecular mass of aspartame?
-
Solve the given system of differential equations by systematic elimination. dx/dt = 2x y dy/dt = x
-
In the 1950s and 1960s the predominant form of merger was the _____________merger. a) horizontal b) vertical c) conglomerate d) diversifying
-
In the late 19th century trusts were formed. They were ;_______________________ the largest trust was the ______________ trust.
-
In 1911 the Supreme Court decided to ________. a) allow the trusts to keep functioning as they had in the past b) break up the trusts c) let the trusts off with small fi nes d) put the leaders of the...
-
Using Gauss elimination and back substitution, solve 8 2 1 4 6 2 3 4 14 X X 3 4 2
-
In 1911 the Supreme Court broke up the______________- and the _______________.
-
Why would the use of insider information be of concern if the market is efficient?
-
Independent random samples of sizes n1 = 30 and n2 = 50 are taken from two normal populations having the means 1 = 78 and 2 = 75 and the variances 21 = 150 and 22 = 200. Use the results of Exercise...
-
Compound A has molecular formula C 5 H 10 . Hydroboration- oxidation of compound A produces 2-methylbutan-1-ol. Draw the structure of compound A: Compound A (C,H10) 1) BH, THF 2) H202, NaOH
-
Explain how the ideal gas law can be deduced for the measurements shown in Figures 1.5 and 1.8. Figure 1.5 Figure 1.8 0.1 L 2.5 2.0 1.5 0.2 L 1.0 0.3 L 0.4 L 0.5 L 0.6 L '0.5 -200 100 0 100 200 300...
-
A bowling ball (a) Rolls across a table (b) Falls on the floor. Is the work associated with each part of this process positive, negative, or zero?
-
Ali is an investor with a high-risk tolerance whose primary investment objective is capital gains. He is a high-income earner, so he does not want to receive current income from his investments....
-
A and B are both retail firms. A sells expensive jewelry and B sells concrete pipes. Both have a June 30 year end. A carries out is stock take on May 30 and B on June 30. With respect to inventory:...
-
Examination of supporting evidence, inquiries of independent persons, and evaluating evidence obtained from other auditing procedures are all examples of Blank______. Multiple choice question....

Study smarter with the SolutionInn App


