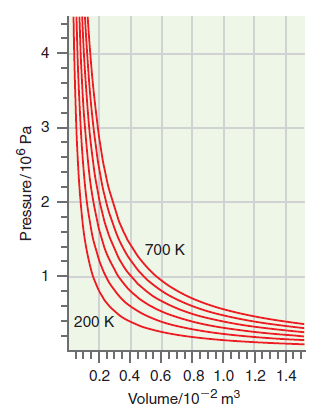
Explain how the ideal gas law can be deduced for the measurements shown in Figures 1.5 and
Question:
Figure 1.5
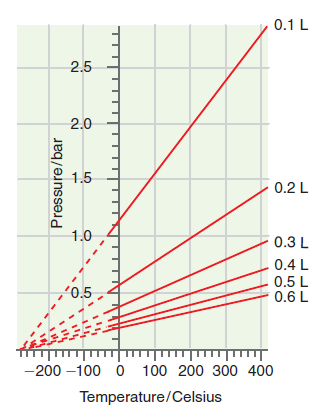
Figure 1.8
Transcribed Image Text:
0.1 L 2.5 2.0 1.5 0.2 L 1.0 0.3 L 0.4 L 0.5 L 0.6 L '0.5 тп -200 –100 0 100 200 300 400 Temperature/Celsius Pressure/bar 700 K 200 K Tпт 0.2 0.4 0.6 0.8 1.0 1.2 1.4 Volume/10-2 m3 3. 2. Pressure/106 Pa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
Figure 15 shows that at constant volume P increases ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Use the ideal gas law to show that, for an ideal gas at constant pressure, the coefficient of volume expansion is equal to = 1/T, where T is the temperature in kelvins. Compare to Table 13-1 for...
-
Pressure of an ideal gas. It is desired to get the pressure exerted by an ideal gas on a wall by accounting for the rate of momentum transfer from the molecules to the wall.? (a) When a molecule...
-
Adiabatic frictionless processes in an ideal gas (a) A gas that obeys the ideal gas law may deviate appreciably from C p = constant. Hence rework Example 11.4-6 using a molar heat capacity expression...
-
CPA firm brings in a yoga instructor during the tax busy season to help relieve stress of the employees. Which is true about the CPA firm's ability to take a deduction for the yoga instructor's...
-
1) What is HiTech's pool rate for the material-handling activity closest to? 2) What is HiTech's pool rate for the automated machinery activity closest to? 3) What is HiTech's pool rate for the...
-
Consider Grassmann variables \(\theta^{\alpha}, \alpha=1,2\), and the even function where 02 = a00. Calculate where D(x, 0) = (x) + 20 a(x) +8 F(x), (28.77) S & (a + a + az), (28.78) d20=- doa doap....
-
What are the characteristics and advantages of the graphical user interface (GUI)?
-
The 2010 balance sheet of Marias Tennis Shop, Inc., showed $680,000 in the common stock account and $4.3 million in the additional paid-in surplus account. The 2011 balance sheet showed $715,000 and...
-
Find the derivative of the function. 54 s(t) = 47 (5t-9)
-
Problem in Financial Analysis Magna Industries Ltd. is being setup to manufacture industrial gears. The expected outlays and proposed financing during construction and the first two operating years...
-
Compound A has molecular formula C 5 H 10 . Hydroboration- oxidation of compound A produces 2-methylbutan-1-ol. Draw the structure of compound A: Compound A (C,H10) 1) BH, THF 2) H202, NaOH
-
A bowling ball (a) Rolls across a table (b) Falls on the floor. Is the work associated with each part of this process positive, negative, or zero?
-
Find the derivative of the function. f (x) = arccot x
-
Global or international recruitment is a straightforward process of hiring employees from anywhere and everywhere, without any geographical barriers. With the popularity of global recruitment,...
-
What are some of the difficulties in providing services in an online environment? What factors differentiate the services sector from the retail sector, for example?
-
y Evaluate 0.3y+ when y = 10 and z = = 5.
-
what are six differences in how expenditures are accrued in governments versus businesses.
-
Can you delineate the role of central banks as key stakeholders in the financial ecosystem, elucidating their functions as monetary authorities responsible for price stability, lender of last resort...
-
Use the circle graph to answer the questions. The Lafayette County Library classifies its DVDs into one of the categories shown below. The library owns 1743 DVDs. Round your answers to the nearest...
-
The graph of an equation is given. (a) Find the intercepts. (b) Indicate whether the graph is symmetric with respect to the x-axis, the y-axis, or the origin. -3 6 -6 3 x
-
At 18C the total volume V of a solution formed from MgS04 and 1.000 kg of water fits the expression v = 1001.21 + 34.69(x - 0.070)2, where v = V/cm3 and x = blb-1. Calculate the partial molar volumes...
-
The following table gives the mole fraction of methylbenzene (A) in liquid and gaseous mixtures with butanone at equilibrium at 303.15 K and the total pressure p. Take the vapour to be perfect and...
-
Use the Gibbs-Duhem equation to show that the partial molar volume (or any partial molar property) of a component B can be obtained if the partial molar volume (or other property) of A is known for...
-
Discuss the key features of the following associated with the financial service industry: Financial products and services of the organisation Financial agency agreements Financial industry codes of...
-
You are building an apartment complex with 80 three-bedroom units near the downtown office district. The designers estimate that there will be on average 3.5 occupants in each apartment. A. Calculate...
-
The field team sends an update to the project manager letting him know that there were 12 employees at the project site, there were no accidents this week and no issues to report, and that the team...

Study smarter with the SolutionInn App


