The standard potential of the half-reaction In a mixture of hydrogen and deuterium, which isotope more favors
Question:
The standard potential of the half-reaction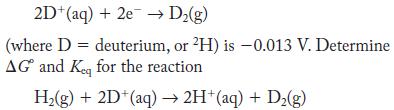
In a mixture of hydrogen and deuterium, which isotope more favors its elemental form under standard conditions?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To determine the Gibbs free energy change G and the equilibrium constant Keq for the reaction stated ...View the full answer

Answered By

Hassan Imtiaz
The following are details of my Professional Experience. Responsibilities Eight years of demanding teaching experience in the field of finance and business studies at Master’s Level. Completion of the given tasks within given time with quality and efficiency. Marketing professional with practical experience in and solid understanding of a diverse range of management applications, including market analysis, sales and marketing, team building and quality assurance. I have excellent skills to approach deal and sustain corporate clients / customers by demonstrating not only extraordinary communication and interpersonal skills but also high caliber presentation, negotiation and closing skills. Manage and follow up the day-to-day activities. Manage and co-ordinate the inventories. Fulfillment of all the tasks assigned.
The following are details of my Areas of Effectiveness. Finance 1. Corporate Finance 2. Advanced Corporate Finance 3. Management of Financial Institutions 4. International Financial Management 5. Investments 6. Fixed Income 7. Real Estate Investment 8. Entrepreneurial Finance 9. Derivatives 10. Alternative Investments 11. Portfolio Management 12. Financial Statement Analysis And Reporting (US GAAP & IFRS) 13. International Financial Markets 14. Public Finance 15. Personal finance 16. Real estate 17. Financial Planning Quantitative Analysis 1. Time Value Of Money 2. Statistics 3. Probability Distribution 4. Business Statistics 5. Statistical Theory and Methods Economics 1. Principles of Economics 2. Economic Theory 3. Microeconomic Principles 4. Macroeconomic Principles 5. International Monetary Economics 6. Money and Banking 7. Financial Economics 8. Population Economics 9. Behavioral Economics International Business 1. Ethics 2. Business Ethics 3. An introduction to business studies 4. Organization & Management 5. Legal Environment of Business 6. Information Systems in Organizations 7. Operations Management 8. Global Business Policies 9. Industrial Organization 10. Business Strategy 11. Information Management and Technology 12. Company Structure and Organizational Management Accounting & Auditing 1. Financial Accounting 2. Managerial Accounting 3. Accounting for strategy implementation 4. Financial accounting 5. Introduction to bookkeeping and accounting Marketing 1. Marketing Management 2. Professional Development Strategies 3. Business Communications 4. Business planning 5. Commerce & Technology Human resource management 1. General Management 2. Conflict management 3. Leadership 4. Organizational Leadership 5. Supply Chain Management 6. Law 7. Corporate Strategy Creative Writing 1. Analytical Reading & Writing Other Expertise 1. Risk Management 2. Entrepreneurship 3. Management science 4. Organizational behavior 5. Project management 6. Financial Analysis, Research & Companies Valuation 7. And any kind of Excel Queries
4.80+
150+ Reviews
230+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Deuterium (D) is the hydrogen isotope of mass number 2, with a proton and a neutron in its nucleus. The chemistry of deuterium is nearly identical to the chemistry of hydrogen, except that the C-D...
-
Deuterium (D) is the isotope of hydrogen of mass number 2, with a proton and a neutron in its nucleus. The chemistry of deuterium is nearly identical to the chemistry of hydrogen, except that the C(D...
-
Deuterium (D, or 2H; is an isotope of hydrogen with atomic mass - 2. Deuterium can be introduced into organic compounds by using reagents in which hydrogen has been replaced by deuterium. Outline...
-
Activity-based costing systems should be used instead of traditional costing systems if ________. A. indirect production costs are a large percentage of production costs B. different products consume...
-
(a) Derive a differential equation for predicting the capsule temperature as a function of time. Solve this equation to obtain the temperature as a function of time in terms of capsule parameters and...
-
Colleges Listed below are the annual tuition amounts of the 10 most expensive colleges in the United States for a recent year. The colleges listed in order are Sarah Lawrence, NYU, George Washington,...
-
Describe situations in which an organization should buy rather than build software components.
-
Lonergan, Inc., a calendar year S corporation in Athens, Georgia, had a balance in AAA of $200,000 and AEP of $110,000 on December 31, 2015. During 2016, Lonergan, Inc., distributes $140,000 to its...
-
Pick any product that is sold in local Canadian market and you are planning to sell it in a country of your choice. Please elaborate on how you will decide on following factors: 1) Adaption method...
-
Disproportionation is a type of redox reaction in which the same species is simultaneously oxidized and reduced. One species that undergoes disproportionation is Cu + (aq).
-
What is G for the oxidation of metallic copper by permanganate (MnO - 4 ) in acidic solution assuming that the copper is oxidized to Cu 2+ ? Use the data in Appendix H.
-
Describe how a brand price elasticity can be calculated from the results of a sales experiment.
-
What is the net force Fnet on an airplane window of area 1400 cm if the pressure inside the cabin is 0.95 atm and the pressure outside is 0.84 atm? Fnet = N
-
Scott also mentioned the following: The expenses for dues and subscriptions were his country club membership dues for the year. $400 of the charitable contributions were made to a political action...
-
One way to measure the strength of a magnetic field is with a flip coil. Suppose a 200 turn, 4.0 cm diameter with a resistance of 2.0 is connected to a 1.0 F capacitor. The coil is held perpendicular...
-
Suppose 8.3 kg of chloroform at 11.4C is poured into 9 kg of propylene glycol at 40.4C. Calculate the final equilibrium temperature (in Celsius), neglecting any energy processes associated with...
-
A long forward contract on a non - dividend - paying stock was entered into some time ago. It currently has six months to maturity. The risk - free rate of interest ( with continuous compounding ) is...
-
What are the audit steps associated with assessing the risk of material misstatement for the revenue cycle?
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
Convert 8720 gal/min to L/min.
-
Table 6.2 lists the range of typical volume flow rates for centrifugal fire-fighting pumps to be 500 to 2500 gal/min. Express this range in the units of ft 3 /s and m 3 /s. Flow rate (m/h) Type of...
-
A certain deep-well pump for a residence is rated to deliver 745 gal/h of water. Express this flow rate in ft 3 /s.
-
What is the amount of overapplied or underapplied overhead Hudson, Inc. has estimated total factory overhead costs of $400,000 and 20,000 direct labor hours for the current fiscal year. If direct...
-
The LLC has assets with a basis of $1,000,000 and a fair market value of $2,400,000. The entity has no liabilities. T has an opportunity to purchase a 25% interest in the entity. The other 75% of the...
-
Five annual payments of $60,000 will be needed to generate a future worth F at an annual interest rate equal to 0.082, calculate the value of F.

Study smarter with the SolutionInn App


