Under certain conditions, biphenyl, C 12 H 10 , can be produced by the decomposition of cyclohexane,
Question:
Under certain conditions, biphenyl, C12H10, can be produced by the decomposition of cyclohexane, C6H12: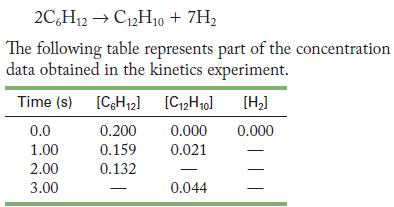
(a) Fill in the missing concentrations.
(b) Calculate the rate of reaction at 1.5 seconds.
Transcribed Image Text:
2C6H12C12H10 + 7H The following table represents part of the concentration data obtained in the kinetics experiment. [C2H0] [H] 0.000 0.000 0.021 Time (s) [C6H12] 0.0 0.200 1.00 0.159 2.00 0.132 3.00 0.044
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The missing concentrations can be calculated using the stoichiometry of the reaction and the given ...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Ethyl alcohol (ethanol) can be produced by the fermentation of sugars derived from trains and other agricultural products. Some countries without large petroleum and natural as reservessuch as...
-
Ethyl alcohol (ethanol) can be produced by the fermentation of sugars derived from agricultural products such as sugarcane and corn. Some countries without large petroleum and natural gas...
-
Oxygen can be produced by the decomposition of potassium chlorate, KClO 3 . The products of the reaction are KCl and O 2 . Write a balanced equation for the reaction.
-
Wilburton Hospital is investigating the possibility of investing in new dialysis equipment. Two local manufacturers of this equipment are being considered as sources of the equipment. After-tax cash...
-
Let T d denote the translation operator (displacement vector d); D (n, ), the rotation operator (n and are the axis and angle of rotation, respectively); and r the parity operator. Which, if any, of...
-
If all of D&R Products other relationships hold, how will Allens worst-case and best-case projections affect the income statement and balance sheet in the first year? D&R Products, Inc., used as an...
-
Given that \(f(x)=\frac{k}{2^{x}}\) is a probability distribution for a random variable that can take on the values \(x=\) \(0,1,2,3\), and 4 , find \(k\).
-
Healthy & Trim Co. offers personal weight reduction consulting services to individuals. After all the accounts have been closed on November 30, 2008, the end of the current fiscal year, the balances...
-
What is the coupon rate for a bond with annual coupons that has a face value of $1,000, 7 years to maturity, a current price of $1,101, and a yield to maturity of 8.4%?
-
For the reaction, the dinitrogen pentoxide appears at a rate of 0.0055 M/s. Calculate the rate at which the NO 2 disappears and the rate of the reaction. (0+)0N (8) + (8)ONZ
-
Dinitrogen tetroxide decomposes to nitrogen dioxide under laboratory conditions. (a) Fill in the missing concentrations. (b) Calculate the rate of reaction at 30 microseconds. NO4(g) 2NO(g) The...
-
A solution contains 0.675 g of ethylamine, C2H5NH2, per 100.0 mL of solution. Electrical conductivity measurements at 20C show that 0.98% of the ethylamine has reacted with water. Write the equation...
-
How do leaders adeptly orchestrate cross-functional synergy, leveraging diverse expertise and perspectives to catalyze innovation and drive transformative initiatives within complex organizational...
-
How do leaders employ sophisticated strategic foresight techniques to anticipate and mitigate emerging challenges within intricate organizational ecosystems?
-
What are the salient features of a quality issue as perceived by consumers and organizations, respectively, delineating the nuanced viewpoints from both ends of the spectrum?
-
What are the primary differences between a secular leader and a servant leader? What is your approach to leadership? Why is diversity an important subject for leadership effectiveness? What can the...
-
What is the relationship between a leaders responsibility for ethical behavior and the idea of an ethical organizational culture? Research a specific nonfictional leader of your choice and provide...
-
Sue now has $490. How much would she have after 8 years if she leaves it invested at 8.5% with annual compounding? a. $941.10 b. $724.64 c. $837.58 d. $1,148.14 e. $1,157.55
-
Willingness to pay as a measure of a person's value for a particular good measures the maximum a person would be willing to pay requires that payment actually be made depends on the satisfaction that...
-
If and determine AB. 6 3] A [4 2. 6 B 2 [5 -1.
-
If and determine AB. A = -5 6. B = [4 6 -5],
-
If and show that (A + B) T = A T + B T . A =| 5 B = 4
-
based on coco cola income statement https://finance.yahoo.com/quote/KO/financials 1. Profit Margins (Gross Profit Margin, EBIT Margin, Net Profit Margin, EBIDTA Margin, EBITDA Growth, Return on...
-
industry comparison of delta airlines and American Airlines financial statement analysis to determine which company you should invest Provide an analysis of the horizontal, common size, and ratio...
-
Modern Electronics Company purchases merchandise inventory from several suppliers. On April 1, 2023, Modern Electronics purchased from Speedy Supplies $120,000 of inventory on account. On May 15,...

Study smarter with the SolutionInn App


