Use the solubility product constant from Appendix F to determine whether a precipitate will form if 10
Question:
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 10 mL 0.0010 M AgNO3 is added to 10 mL 0.0010 M Na2SO4.
Appendix F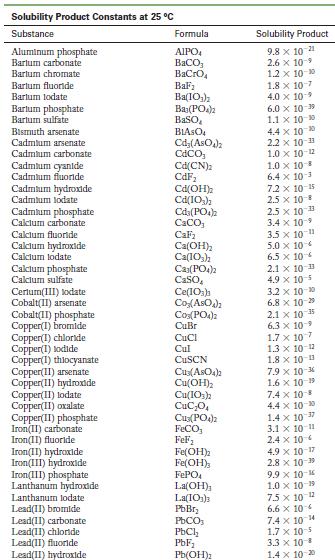

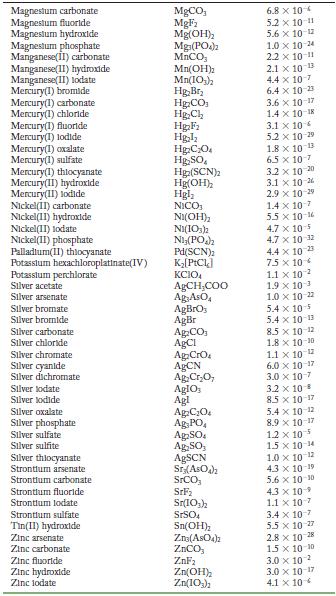
Transcribed Image Text:
Solubility Product Constants at 25 C Substance Aluminum phosphate Bartum carbonate Bartum chromate Barlum fluoride Barlum lodate Bartum phosphate Bartum sulfate Bismuth arsenate Cadmium arsenate Cadmium carbonate Cadmium cyanide Cadmium fluoride Cadmium hydroxide Cadmium lodate Cadmium phosphate Calcium carbonate Calcium fluoride Calcium hydroxide Calcium lodate Calcium phosphate Calcium sulfate Cerlum(III) lodate Cobalt(II) arsenate Cobalt(II) phosphate Copper(1) bromide Copper (1) chloride Copper (1) todide Copper(1) thiocyanate Copper(11) arsenate Copper(II) hydroxide Copper(II) lodate Copper(II)oxalate Copper(II) phosphate Iron(II) carbonate Iron(11) fluoride Iron(II) hydroxide Iron(III) hydroxide Iron(III) phosphate Lanthanum hydroxide Lanthanum lodate Lead(11) bromide Lead(11) carbonate Lead(11) chloride Lead(11) fluoride Lead(II) hydroxide Formula AIPO BaCO3 BaCrO BaF Ba(10) Ba3(PO4)2 BaSO BIASO Cd(AsO4)2 CdCO Cd(CN) CdF Cd(OH) Cd(103) Cd3(PO4)2 CaCO, CaF Ca(OH) Ca(10) C23(PO4)2 CaSO Ce(103)1 Co(ASO) CO3(PO4)2 CuBr CuCl Cul CuSCN Cu3(ASO4)2 Cu(OH) Cu(103)2 CuC0 Cu3(PO4)2 FeCO FeF Fe(OH)2 Fe(OH)3 FePO4 La(OH) La(IO3)3 PbBr PbCO PbCl PbF Pb(OH) Solubility Product 9.8 x 10 21 2.6 x 10-9 1.2 x 10-10 1.8 x 10-7 4.0 x 109 6.0 x 10-19 1.1 x 10-10 4.4 x 100 2.2 x 10-11 1.0 x 10-12 1.0 x 10-8 6.4 x 10- 7.2 x 10-15 2.5 x 10-8 2.5 x 10-3 3.4 x 10 3.5 x 10 11 5.0 x 10 6.5 x 10 2.1 x 10 11 4.9 x 10-5 3.2 x 10-20 6.8 x 10-29 10-1 2.1 x 6.3 10- 1.7 x 107 1.3 x 10-12 1.8 x 10-13 7.9 x 10-6 1.6 x 10-19 7.4 x 10-8 4.4 x 10-0 1.4 x 10-7 3.1 x 10-11 2.4 x 10 4.9 x 10-17 2.8 x 10-9 9.9 x 10- 1.0 x 10-19 75 x 10-2 6.6 x 10 7.4 x 10 4 1.7 x 10-5 3.3 x 10 B 1.4 x 10-20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Q 12 10 ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 25.0 mL of 0.010 M NaOH is added to 75.0 mL of a 0.10 M solution of magnesium chloride? Appendix F...
-
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 10.0 mL of 1.0 10 -6 M iron(II) chloride is added to 20.0 mL of 3.0 10 -4 M barium hydroxide....
-
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 20.0 mL of 1.0 10 -6 M magnesium chloride is added to 80.0 mL of 1.0 10 -6 M potassium fluoride....
-
Write the complete APT part program to profile mill the outside edges of the part. The part is 15 mm thick. Tooling = 30 mm diameter end mill with four teeth, cutting speed = 150 mm/min, and feed =...
-
The main trouble with variable costing is that it ignores the increasing importance of fixed costs in manufacturing companies? Do you agree? Why?
-
According to the Ameriprise Financial Money Across Generations study, 9 out of 10 parents with adult children ages 20 to 35 have helped their adult children with some type of financial assistance...
-
Evaluate machine XYZ on the basis of the PW method when the MARR is $12 \%$ per year. Pertinent cost data are as follows: Investment cost Useful life Market value Annual operating cost Overhaul cost...
-
Evan Neal operates a roller skating center. He has just received the monthly bank statement at January 31 from Citizen National Bank, and the statement shows an ending balance of $575. Listed on the...
-
ces The most recent financial statements for Crosby, Incorporated, follow. Sales for 2021 are projected to grow by 25 percent. Interest expense will remain constant; the tax rate and the dividend...
-
Calculate the solubility of copper(II) iodate, Cu(IO 3 ) 2 (K sp = 7.4 10 -8 ), in (a) Water. (b) A 0.10 M copper(II) nitrate solution.
-
Calculate the solubility of barium sulfate (K sp 1.1 10 -10 ) in (a) Water. (b) A 0.10 M barium chloride solution.
-
If i = 12%, what is the value of B in the diagram? 800 s00 800 1.5B
-
Under the cash basis of accounting assurance-type warranty costs are charged to operating expenses ____________________. Select answer from the options below when warranty claims are certain based on...
-
explain the origin, purpose, and description of DRGs. Include what payment is based on. Identify the benefits and problems with reimbursement via this method. Explain how you as a nurse-manager can...
-
Dow Chemical CEO, Andrew Liveris, checks financial reports to see whether Dow has met its financial goals for the quarter.
-
Indicate how each of the following items is reported on the partnership tax return. What items are reported as a portion of Ordinary Income and what items are Separately Stated? Dividend Income...
-
What is the gross profit (GP) of the original bill? (Sale price less COGS = GP) Food: COGs is 30% therefore the remaining profit is 70% of the food cost (food cost X 0.7) Wine: COGs is 20% therefore...
-
Demonstrate that an at-the-money call option on a given stock must cost more than an at-the money put option on that stock with the same maturity. The stock will pay no dividends until after the...
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
If air has a constant specific weight of 0.075 lb/ft 3 , what pressure difference would result when driving from the base to the top of Pikes Peak, if the climb for the trip is 8400 ft?
-
Figure 4.21 shows a vacuum tank with a flat circular observation window in one end. If the pressure in the tank is 0.12 psia when the barometer reads 30.5 in of mercury, calculate the total force on...
-
The flat left end of the tank shown in Fig. 4.21 is secured with a bolted flange. If the inside diameter of the tank is 30 in and the internal pressure is raised to +14.4 psig, calculate the total...
-
Assume convergence of yields to the mid-point of 3.61% and 3.26%, what is the profit for a successful trading where the long position is a $1,000 face value bond?
-
In addition to using the course textbook, conduct additional online research this week and write an essay that explains the following: What are currency futures contracts? Why do companies enter into...
-
What happens if a company does not pay dividends? What if dividends are not a priority? How do you evaluate them?

Study smarter with the SolutionInn App


