Question: Which elements can form expanded valence shell molecules? Do the two xenon compounds shown below have an expanded valence shell? Explain your answer. XeF2 XeF4
Which elements can form expanded valence shell molecules? Do the two xenon compounds shown below have an expanded valence shell? Explain your answer.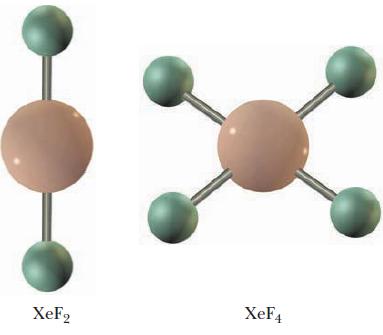
XeF2 XeF4
Step by Step Solution
3.38 Rating (148 Votes )
There are 3 Steps involved in it
The two xenon compounds shown in the image XeF2 and XeF4 do not have expanded valence shells The oct... View full answer

Get step-by-step solutions from verified subject matter experts


