Write balanced equations for the following reactions. (a) NH4 + NO4N + HO (b) F + HO
Question:
Write balanced equations for the following reactions.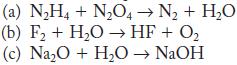
Transcribed Image Text:
(a) N₂H4 + N₂O4N₂ + H₂O (b) F₂ + H₂O → HF + O₂ (c) Na₂O + H₂O → NaOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a 2N 2 H 4 N 2 O ...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write balanced nuclear equations for the following reactions and identify X:
-
Write balanced nuclear equations for the following reactions and identify X: 80 34
-
Write balanced equations for each of the following reactions (some of these are analogous to reactions shown in the chapter). (a) Aluminum metal reacts with acids to form hydrogen gas. (b) Steam...
-
Data for Barry Computer Co. and its industry averages follow. a. Calculate the indicated ratios for Barry. b. Construct the DuPont equation for both Barry and the industry. c. Outline Barrys...
-
A lossless transmission line with a characteristic impedance of 75 is terminated by a load of 120. The length of the line is 1.25. If the line is energized by a source of 100 V (rms) with an internal...
-
a) Sketch the graph of y = g(x), where g(x) = 2 x . b) On separate diagrams, sketch the graphs of i) y = g(x + 2) ii) y = g(x) 1 iii) y = g(x) + 4 Mark on each sketch, where possible, the...
-
According to an article in the Wall Street Journal, in 2022, the Walt Disney Company hired 50 people for a new division of the company to develop metaverse strategiesfinding ways to tell interactive...
-
A survey asks respondents to respond to the statement My work is interesting. Interpret the frequency distribution shown here (taken from an SPSS output): My work isinteresting: Category Abs. Freq...
-
The formula of a compound is Y3(PO4)2. The electronic configuration of the atom of Y is 15. A. 2.8.2 B. 2.8.3 C 2.8.4 16. D. 2.8.5 The atomic numbers and mass numbers of atoms, W, X, Y and Z are...
-
A national catalog and Internet retailer has three warehouses and three major distribution centers located around the country. Normally, items are shipped directly from the warehouses to the...
-
Balance these reactions. (a) Al(s) + O(g) AlO3(s) (b) N(g) + H(g) NH3(g) (c) CH%(l) + Oz(g) H,O(l) + CO,(g)
-
In an experiment performed in the laboratory, 44 g NH 3 is mixed with 120 g O 2 , and 73 g NO is isolated. Given the following equation, what is the percent yield? 4NH3 + 50 4NO + 6HO
-
The 2021 income statement of Adrian Express reports sales of $19,310,000, cost of goods sold of $12,250,000, and net income of $1,700,000. Balance sheet information is provided in the following...
-
Anna is running to the right, as shown in Figure Q3.19. Balls 1 and 2 are thrown toward her by friends standing on the ground. According to Anna, both balls are approaching her at the same speed....
-
A, B, and C are three similar plants under the same management who wants to merge them for better operation. The details are as under: You have to find out: (i) the capacity of the merged plant for...
-
For the DOS, we are interested in how MS and gender are related with the depression outcome. Based on the logistic model: Dep \(\sim\) MS | Gender (MS, Gender, and their interaction), answer the...
-
A couple nearing retirement wishes to purchase a new home costing \($750\),000, make a 20 percent down payment, and finance the remaining \($600\),000. The conventional 15-year loan rate is 5.6250...
-
Identify the sample.
-
Meyer Inc's assets are $745,000, and its total debt outstanding is $185,000. The new CFO wants to establish a debt ratio of 55%. The size of the firm does not change. How much debt must the company...
-
Following is the current balance sheet for a local partnership of doctors: The following questions represent independent situations: a. E is going to invest enough money in this partnership to...
-
Predict the change in the partial pressure of CO2 as Xe gas is introduced into the reaction vessel at constant volume and temperature. CO(g) + 1/2O 2 (g) CO 2 (g) at equilibrium for which H o R =...
-
You place 3.00 mol of NOCl(g) in a reaction vessel. Equilibrium is established with respect to the decomposition reaction NOCl(g) NO(g) + 1/2Cl 2 (g). a. Derive an expression for K P in terms of the...
-
Predict the change in the partial pressure of CO2 as a platinum catalyst is introduced into the reaction vessel at constant volume and temperature. CO(g) + 1/2O 2 (g) CO 2 (g) at equilibrium for...
-
In the Depreciation Recapture Example, if Mike sells the property for $350,000, how much income would he realize?
-
What impact do leadership styles and behaviors have on team-building outcomes, and how can leaders adapt their approach to leadership to accommodate diverse team compositions, work contexts, and...
-
= On 1/1/X4, Phillip invested $1,100,000 in Sleeper's ordinary shares (35% owned). Sleeper reported: Assets $3,500,000 Liabilities 600,000 The book value of Sleeper's net assets approximated market...

Study smarter with the SolutionInn App


