Write the rate law and the molecularity for each of the following elementary reactions. (a) NO+NOCl NO
Question:
Write the rate law and the molecularity for each of the following elementary reactions.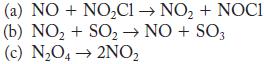
Transcribed Image Text:
(a) NO+NOCl NO + NOCI (b) NO + SO NO + SO3 (c) NO4 2NO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The rate law for an elementary reaction can be written directly from the reactions stoichiometry bec...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write the expected rate law and molecularity for each of the following elementary reactions in the gas phase. Strategy The rate law for an elementary reaction is derived directly from its...
-
What is the molecularity of each of the following elementary reactions? Write the rate law for each. (a) 2 NO(g) N2O2(g) (c) SO3(g) SO2(g) + O(g)? CH2 H2C-CH2(g)- CH2_ CH-CH3(g)
-
Write the rate law and the molecularity for each of the following elementary reactions. (a) CH5C1 CH4 + HC1 (b) NO + O3 NO + O (c) HI + CHI CH6 + 1
-
One of the key aspects of the Risk Management Process which the Health Care Providers should be using as an essential tool is risk modeling. It can be argued that for the sustainability of the Health...
-
Given the circuit diagram in figure, find the following voltages: Vda, Vbh, Vgc, Vfa, Vac, Vai, Vhf, and Vdc, 8 V 12 V 20 V 8 V 14 V 16 V
-
How external, external-internal, and internal documentary evidence are generally defined?
-
Table B. 22 contains data on 1916 team performance for Major League Baseball. Use all possible regressions to build a model for this data. Perform a residual analysis on the final model and comment...
-
On November 1, 2022, the account balances of Hamm Equipment Repair were asfollows. During November, the following summary transactions were completed.Nov. 2 Paid $700 for salaries due employees.10...
-
Consider an agent with the following utility function: U = E(rp) 0.005A Consider that this agent wants to create an efficient portfolio and that her coefficient of risk aversion is 25. Compute the...
-
Nitrogen dioxide reacts with carbon monoxide to form carbon dioxide and nitrogen monoxide. NO(g) + CO(g) CO(g) + NO(g) Two mechanisms are proposed: Mechanism I (one step): NO(g) + CO(g) CO(g) + NO(g)...
-
Evaluate each of the following proposed mechanisms to determine whether it is consistent with the experimentally determined stochiometry and rate law, and identify intermediates, if any. 2NO2+O3 NO5...
-
A firm issues long-term debt with an effective interest rate of 10%, and the proceeds of this debt issue can be invested to earn an ROI of 12%. What effect will this financial leverage have on the...
-
1. Gas phase decomposition of di-t-butyl peroxide is monitored in a batch reactor of constant volume at isothermal conditions of 170C. (CH3)3COOC(CH3)3 C2H6+2 CH3COCH3 The run is started with pure...
-
What is the correlation for following? A. Organiation commitment to Job satisfaction? Affective Commitment to Job Satisfaction? Continuance COmmitment to Job Satisfaction? Normative Commitment to Job...
-
Explore the factors related to job satisfaction, and Review literature to define job satisfaction, Write down an event or stories related to job satisfaction, within any organisation organisation or...
-
Questions: Describe the concept of value as it relates to value analysis. Provide examples of how an organization can increase value to itself or to its customers. Questions :In general, do you...
-
Analyze the assigned Company's Operations and Logistics practices: a) Explain if the company's supply chain strategy is a "market responsive" or "physically efficient" Supply Chain Strategy? Why do...
-
Effect of transactions on various financial ratios. Include the effect that each transaction/event listed here will have on the financial ratio listed opposite it, and provide an explanation for your...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Determine the horizontal displacement of joint D. Assume the members are pin connected at their end points. AE is constant. Use the method of virtual work. 8 m 2k D. 6 ft 3k 6 ft
-
Determine the vertical displacement of joint A.The cross-sectional area of each member is indicated in the figure. Assume the members are pin connected at their end points.E = 29 (10) 3 ksi. Using...
-
Determine the vertical displacement of joint A.The cross-sectional area of each member is indicated in the figure. Assume the members are pin connected at their end points. E = 29 (10) 3 ksi. Use the...
-
Given the following method: 01 public static double median (double[] d) { double median = Double. NaN; 02 03 04 if (d != null && d.length > 0) if (d.length == 1) { median = d[0]; } else {...
-
What is the minimum number of test cases needed to meet statement coverage criterion for the following code? int total; public int show(int val) { total = 0; if (val <0)( } val = -val; while (val >...
-
AreaVicki owns and operates a baby food manufacturing business (as a sole proprietorship). During 2023, she incurred expenses of 240,000 in research and experimental expenditures. For regular tax...

Study smarter with the SolutionInn App


