A laser pointer used in a lecture hall emits light at 650 nm. What is the frequency
Question:
A laser pointer used in a lecture hall emits light at 650 nm. What is the frequency of this radiation? Using Figure 6.4, predict the color associated with this wavelength.
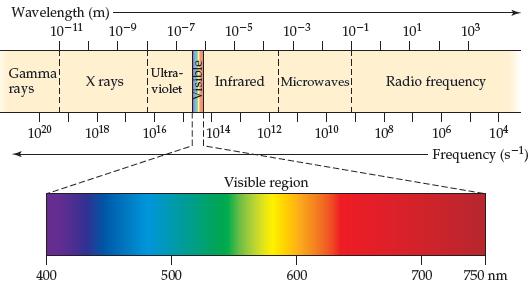
Wavelength (m)- 10-9 10-11 10-7 10-5 10-3 10-1 101 103 Gamma! X rays !Ultra- i violet rays Infrared iMicrowavesi Radio frequency 1020 1018 1016 1014 1012 1010 108 106 104 Frequency (s-1) Visible region 400 500 600 700 750 nm
Step by Step Answer:
The wavelength of a laser is 650 nm 650 10 ...View the full answer



Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Related Video
Using a few basic physics principles, you can impress your friends with a trick that makes a bottle disappear. For this experiment, you will need a mini plastic bottle, glycerin, a glass, and a funnel. First, open the bottle and pour some glycerin into it, then close the bottle tightly. Next, pour some water into the glass using a funnel. Place the mini bottle into the glass of water and it will look normal. This is because light travels through air faster than it travels through the glass and water, allowing our eyes to see the bottle inside the glass. However, when you fill the mini bottle with more glycerin, and pour glycerin into the glass, then put the glycerin filled bottle into the glass with glycerin in it. Half of the bottle that is submerged in the glycerin will become invisible as the light travels through glass and glycerin at the same speed, thus it does not bend and no refraction takes place, making the bottle invisible. This happens because both glass and glycerin have almost the same refractive index, which causes the speed of light to be the same in both mediums, causing no bending of light and making the bottle disappear.
Students also viewed these Sciences questions
-
(a) A red laser pointer emits light with a wavelength of 650 nm. What is the frequency of this light? (b) What is the energy of one of these photons? (c) The laser pointer emits light because...
-
(a) A green laser pointer emits light with a wavelength of 532 nm. What is the frequency of this light? (b) What is theenergy of one of these photons? (c) The laser pointer emits light because...
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
Determine the force in member GC of the truss and state if this member is in tension or compression. Units Used: kip = 103 lb Given: F1 = 1000 lb F2 = 1000 lb F3 = 1000 lb F4 = 1000 lb a = 10 ft =...
-
y varies jointly as x and z and inversely as the square of w, and y = 12 / 5 when x = 16, z = 3, and w = 5 Find an equation of variation for the given situation.
-
An engine causes a car to move 10 meters with a force of 100 N. The engine produces 10,000 J of energy. What is the efficiency of this engine?
-
Visit www.pearsonglobaleditions.com/malhotra to read the video case and view the accompanying video. Marriott: Marketing Research Leads to Expanded Offerings highlights Marriott's discovery from...
-
An inexperienced accountant for Nerys Corporation showed the following in the income statement: income before income taxes and extraordinary item $400,000, and extraordinary loss from flood (before...
-
W Nov. 8 8 11 11 13 14 16 Natalie cashes her U.S. Savings Bonds and receives $630, which she deposits in her personal bank account. She opens a bank account under the name "Cookie Creations" and...
-
The following narrative describes the processing of customer mail orders at Eye- Dee- A- Pet, Inc.: Eye- Dee- A- Pet, Inc. is a small manufacturing operation engaged in the selling of digital...
-
The energy from radiation can be used to cause the rupture of chemical bonds. A minimum energy of 242 kJ/mol is required to break the chlorinechlorine bond in Cl 2 . What is the longest wavelength of...
-
(a) Consider the following three statements: (i) A hydrogen atom in the n = 3 state can emit light at only two specific wavelengths, (ii) A hydrogen atom in the n = 2 state is at a lower energy than...
-
An alternative form of estimation is accomplished through the method of moments. The method involves equating the population mean and variance to the corresponding sample mean x and sample variance s...
-
What is the Yield to Maturity (YTM) on a 3.65% T-Bond with a maturity date in October 2021 if the current market price is 111.82? How should you interpret this YTM? A) YTM = 0.46%, the maximum...
-
24. X Corp. issues a bond on July 1, 2005 with a maturity date of July 1, 2019. The bond has a 5% coupon and was issued for par. Its stated redemption price and issue price was $10,000 per bond. On...
-
Practice focus groups are particularly used for which of these? Obtaining firsthand information about relatives of employees. Addressing issues when they get out of hand. Obtaining firsthand...
-
The inventory of XYZ Company on November 30, 2014 shows 2500 units at 12,5 per unit. Revenue from sales for December totals 64,000 (= 3,200 units at 20 per unit). The following purchases were made...
-
Mr. E. Kisan is eager to make his utility function explicit. So he approaches his financial consultant, Mr. Chelumala Rishi Revanth for help who places before Mr. Kisan a choice of a lottery giving...
-
Comprehensive The following are Farell Corporation's balance sheets of as December 31, 2013, and 2012, and the statement of income and retained earnings for the year ended December 31, 2013:...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Three solutions are mixed together to form a single solution. One contains 0.2 mol Pb(CH3COO)2, the second contains 0.1 mol Na2S, and the third contains 0.1 mol CaCl2. (a) Write the net ionic...
-
Which of the following solutions has the largest concentration of solvated protons: (a) 0.2 M LiOH (b) 0.2 M HI (c) 1.0 M methyl alcohol (CH3OH)? Explain
-
Use the molecular representations shown here to classify each compound as either a nonelectrolyte, a weak electrolyte, or a strong electrolyte (see inside back cover for element color scheme). (a)...
-
Blair, CPA, uses the cash receipts and disbursements method of reporting. In Year 7, a client gave Blair 100 shares of a listed corporation's stock in full satisfaction of a $5,000 accounting fee the...
-
What is driving the increase in the price of healthcare services today? Explain why it is important to know the scope of business being reviewed when using financial statements. Explain the content...
-
What is a discount? How do you activate discounts? Activate discounts: Assign a discount account: 3. Discounts Given - What is the account type and detail type for this account?

Study smarter with the SolutionInn App


