A glass capillary tube with a radius of 0.050 mm is inserted into an open container filled
Question:
A glass capillary tube with a radius of 0.050 mm is inserted into an open container filled with an unknown liquid, and the meniscus has an appearance similar to Figure 10.35A. It is found that the surface tension causes the liquid in the tube to rise a distance of 2.5 cm relative to the surface of the liquid outside the tube. If the density of the liquid is 1000 kg/m1what is the surface tension?
Figure 10.35A
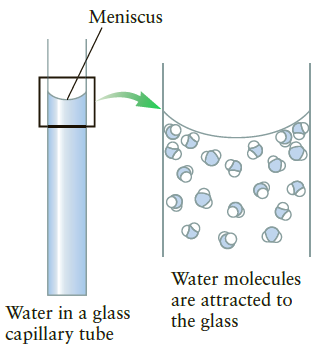
Meniscus Water molecules are attracted to the glass Water in a glass capillary tube
Step by Step Answer:
The pressure on the liquid inside is the capillary pressur...View the full answer



College Physics Reasoning and Relationships
ISBN: 978-0840058195
2nd edition
Authors: Nicholas Giordano
Related Video
The property of the surface of a liquid that allows it to resist an external force, due to the cohesive nature of its molecules. The cohesive forces between liquid molecules are responsible for the phenomenon known as surface tension. The molecules at the surface of a glass of water do not have other water molecules on all sides of them and consequently, they cohere more strongly to those directly associated with them (in this case, next to and below them, but not above). is not really true that a \\\"skin\\\" forms on the water surface; the stronger cohesion between the water molecules as opposed to the attraction of the water molecules to the air makes it more difficult to move an object through the surface than to move it when it is completely submersed
Students also viewed these Sciences questions
-
A glass capillary tube with a diameter of 0.50 mm and length 10 cm is filled with a salt solution with a resistivity of 0.10m. What is the resistance?
-
Figure P6.6 shows an angled, glass capillary tube with diameter 110 m. Other dimensions are shown. (a) Where will the top of the capillary rise be? (b) What is the water pressure in the horizontal...
-
The contact angle for water on clean glass is close to zero. Calculate the surface tension of water at 30C given that at that temperature water climbs to a height of 9.11 cm in a clean glass...
-
Alleghany Community College operates four departments. The square footage used by each department is shown below. Alleghany's annual building rental cost is $320,000 What amount of rent expense that...
-
Prepare an amortization schedule for a three-year loan of $75,000. The interest rate is 8 percent per year, and the loan calls for equal annual payments. How much interest is paid in the third year?...
-
In Problems 58, determine whether the given vectors u and v are linearly dependent or linearly independent. u = (0 , 2), v = (3 , 0)
-
The pumper truck shown in Fig. P5.107 is to deliver \(1.5 \mathrm{ft}^{3} / \mathrm{s}\) to a maximum elevation of \(60 \mathrm{ft}\) above the hydrant. The pressure at the 4-in.-diameter outlet of...
-
Hugh Leach Corp., a producer of machine tools, wants to move to a larger site. Two alternative locations have been identified: Bonham and McKinney. Bonham would have fixed costs of $800,000 per year...
-
Please summarize the changing use of positive psychology over time, including how it compares with other theories in personality psychology. Consider the future relevance of positive psychology...
-
Read the case study and answer the question below with a one page response. What does a SWOT analysis reveal about the overall attractiveness of Under Armours situation? Founded in 1996 by former...
-
Repeat Problem 72, but now for a tube of radius 10 nm. Express your answer in pascals and as a ratio relative to atmospheric pressure. Data from Problem 72 What is the capillary pressure for water in...
-
The viscosity of normal (healthy) blood is about three times greater than the viscosity of water. Certain diseases such as polycythemia can cause the viscosity of blood to be as much as three times...
-
On January 1, 2024, Rapid Airlines issued $200 million of its 8% bonds for $184 million. The bonds were priced to yield 10%. Interest is payable semiannually on June 30 and December 31. Rapid...
-
What is the effect of a viscosity (competence) difference between strain markers and the matrix?
-
If the elasticity of aggregate cigarette demand with respect to advertising were 0.15 in absolute value, by extrapolation what effect on cigarette consumption would be caused by a 10 percent...
-
How can extensional faults form in a thrust-and-fold belt?
-
What types of materials are truly viscous? What parts of the Earth can be modeled as being viscous?
-
What is the difference between shear fractures and faults?
-
Identify the auditing firm that audited the financial statements of WestJet in Appendix III.
-
Ask students to outline the reasons why the various elements of culture (social structures and control systems, language and aesthetics, religion and other belief systems, educational systems, etc.)...
-
Are the effective nuclear charges listed in Figure 21.13 helpful in explaining the trend in the electron affinity with increasing atomic number? Explain your answer. 1s 1 1s 1.69 Li 2s 1.28 2s 1.91...
-
Are the effective nuclear charges listed in Figure 21.13 helpful in explaining the trend in the first ionization energy with increasing atomic number? Explain your answer. 1s 1 1s 1.69 Li 2s 1.28...
-
The electron affinities of He, Be, and Ne are negative, meaning that the negative ion is less stable than the neutral atom. Explain why this is so for these three elements.
-
Excerpts from Andre Company's December 31, 2024 and 2023, financial statements are presented below: Accounts receivable Inventory Net sales Cost of goods sold Total assets Net income Total...
-
The York City Hospital has just acquired new equipment. The equipment cost $ 4 , 2 5 0 , 0 0 0 , and the organization spent $ 1 3 5 , 0 0 0 on upgrading the physical plant to the new equipment will...
-
Carmen Camry operates a consulting firm called Help Today, which began operations on December 1. On December 31, the company's records show the following selected accounts and amounts for the month...

Study smarter with the SolutionInn App


