Question: Which choice correctly identifies the three processes shown in the diagrams? (a) I = isobaric; II = isochoric; III = adiabatic (b) I = isothermal;
Which choice correctly identifies the three processes shown in the diagrams?
(a) I = isobaric; II = isochoric; III = adiabatic
(b) I = isothermal; II = isothermal; III = isobaric
(c) I = isochoric; II = adiabatic; III = isobaric
(d) I = isobaric; II = isothermal; III = isochoric
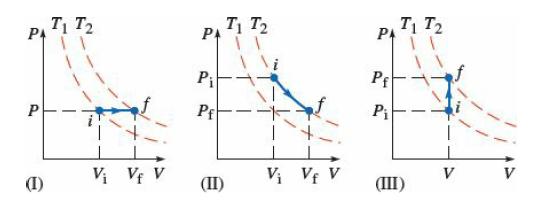
T T2 PA T T2 PA PA T1 T2 Pi Pf P Pf P V; VE V V; Vf V V V (1) (II) (III)
Step by Step Solution
3.42 Rating (171 Votes )
There are 3 Steps involved in it
c I isochoric II adiabatic III isobaric Explanation Isochoric process A proc... View full answer

Get step-by-step solutions from verified subject matter experts


