Which of the following diagrams best represents the size of the atomic nucleus relative to the size
Question:
Which of the following diagrams best represents the size of the atomic nucleus relative to the size of the atom?
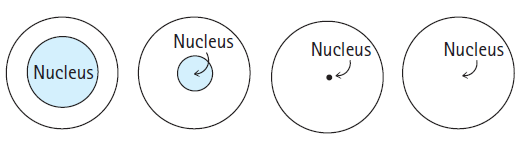
Transcribed Image Text:
Nucleus Nucleus Nucleus Nucleus)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (22 reviews)
The one on ...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
Which of the following diagrams best represents an aqueous solution of NaF? The water molecules are not shown for clarity. Will this solution be acidic, neutral, or basic?
-
Which of the following diagrams best represents a strong acid, such as HCl, dissolved in water? Which represents a weak acid? Which represents a very weak acid? (The hydrated proton is shown as a...
-
A gaseous sample of a substance is cooled at constant pressure. Which of the following diagrams best represents the situation if the final temperature is (a) Above the boiling point of the substance...
-
A potential difference of 1.20 V will be applied to a 33.0 m length of 18-gauge copper wire (diameter = 0.0400 in.). Calculate (a) The current, (b) The magnitude of the current density, (c) The...
-
The Davidson Corporation's balance sheet and income statement are provided here. Davidson Corporation: Income Statement for Year Ending December 31, 2012 (Millions of Dollars)...
-
(a) Arrange the chlorine oxoacids in order of increasing oxidizing strength. (b) Suggest an interpretation of that order in terms of oxidation numbers.
-
The linkage is made of two pin-connected A-36 steel members, each having a cross-sectional area of 1.5 in 2 . Determine the magnitude of the force P needed to displace point A 0.025 in. downward. 2...
-
Refer to the preceding facts for Pontiacs acquisition of 80% of Starks common stock and the bond transactions. Pontiac uses the simple equity method to account for its investment in Stark. On January...
-
Selected cost data for Aerial Co for the year are as follows: Estimated overhead cost for the year Estimated direct labor cost for the year Actual manufacturing overhead cost for the year Actual...
-
Gauntlet Gyms Inc. owns athletic training facilities and camping grounds, which it leases to corporate clients, educational institutions, and other non-governmental organizations. The Xavier...
-
As a tree respires, it takes in carbon dioxide, CO 2 , and water vapor, H 2 O, from the air, while also releasing oxygen, O 2 . Does the tree lose or gain weight as it respires? Explain.
-
A beam of protons and a beam of neutrons of the same energy are both harmful to living tissue. The beam of neutrons, however, is less harmful. Suggest why.
-
Fessler Enterprises, Inc., lost its entire inventory in a hurricane that occurred on May 31, 2016. Over the past five years, gross profit has averaged 39 percent of net sales. The company's records...
-
Suppose most fans prefer Sunday afternoon baseball games (regardless of opponent) to all other types of games. Describe two pricing strategies that a team could use to increase profits based on this...
-
Should Pudge Buffet stay in the farming business?
-
You are the commissioner of the National Hockey League. You have been called to testify at an antitrust case against the NHL. Argue that a. The NHLis not a monopoly. b. Even if it is a monopoly, it...
-
How much waste does a price ceiling on heating oil cause?
-
How do cost-saving innovations affect economic profit in the short run? In the long run?
-
Jim Culf is the manager of the Royal Hotel, Bigtown-on-Sea. In anticipation of preparing next years budget, he has analysed his recent costs and income. His findings are summarized below....
-
If someone's Z-score for a variable was 0.67. Their score is a significant extreme score. Their score is not significant. O Their score is slightly above average. O Their score is an outlier.
-
In cylindrical coordinates, let = 0 for < 1 mm, = 2 sin(2000 ) nC/m 3 for 1 mm < < 1.5 mm, and = 0 for > 1.5 mm. Find D everywhere.
-
The line segment x = 0,1 y 1, z = 1, carries a linear charge density L = |y| C/m. Let z = 0 be a conducting plane and determine the surface charge density at: (a) (0, 0, 0); (b) (0, 1, 0).
-
Atomic hydrogen contains 5.5 10 25 atoms/m 3 at a certain temperature and pressure. When an electric field of 4 kV/m is applied, each dipole formed by the electron and positive nucleus has an...
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


