Vapor pressure data for chlorine are given below. a. Use these data and the ClausiusClapeyron equation (Equation
Question:
Vapor pressure data for chlorine are given below.
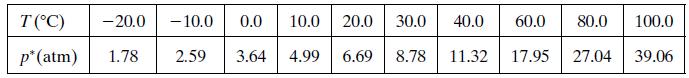
a. Use these data and the Clausius–Clapeyron equation (Equation 6.1-3) to estimate the heat of vaporization of chlorine (kJ/mol) and to obtain an expression for P*Cl2 (T).
b. What is the operating pressure in the chlorine vaporizer (torr)?
c. At what rate (kW) must heat be added to the chlorine in the vaporizer?
Transcribed Image Text:
T(°C) - 20.0 - 10.0 0.0 10.0 20.0 30.0 40.0 60.0 80.0 100.0 p*(atm) 1.78 2.59 3.64 4.99 6.69 8.78 11.32 17.95 27.04 39.06
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (15 reviews)
a The ClausiusClapeyron equation is given by dPdT HvapRT2 where Hvap is the heat of vaporization of ...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Question Posted:
Students also viewed these Business questions
-
Estimate the heat of vaporization (kJ/mol) of benzene at a pressure of 100 mm Hg, using each of the following correlations and data: (a) The heat of vaporization at the normal boiling point given in...
-
Estimate the heat of vaporization (kJ/mol) of benzene at 25C, using each of the following correlations and data: (a) The heat of vaporization at the normal boiling point and Watsons correlation. (b)...
-
Estimate the heat of vaporization of ethyl benzene at its normal boiling point using Troutons rule and Chens rule and compare the results with a tabulated value of this quantity. Then estimate Hv at...
-
Calculate the 90% confidence interval for the difference (mu1-mu2) of two population means given the following sampling results. Population 1: sample size = 19, sample mean = 20.52, sample standard...
-
(a) How is a reaction quotient used to determine whether a system is at equilibrium? (b) If Qc > Kc, how must the reaction proceed to reach equilibrium? (c) At the start of a certain reaction, only...
-
Marwick s Pianos, Incorporated, purchases pianos from a large manufacturer for an average cost of $ 1 , 5 0 3 per unit and then sells them to retail customers for an average price of $ 3 , 3 0 0...
-
Consider a family of call options on a non-dividend-paying stock, each option being identical except for its strike price. The value of the call with strike price \(K\) is denoted by \(C(K)\). Prove...
-
A company is trying to determine the proper capacity level for its new electric car. A unit of capacity provides the potential to produce one car per year. It costs $10,000 to build a unit of...
-
need help to mark my working and answer and provide explanation if wrong Image transcription text A power plant operates with a high-pressure steam of 50 bar and has a boiler exit temperature of 600...
-
What are the four management functions?
-
The plant is to produce 3.5 10 6 kg/year of "67% Cl." Assuming that the plant operates 300 days per year, 24 hours per day, calculate the required hourly feed rates (kg/h) of the 10 wt% PVC slurry...
-
Vapor pressure data for chlorine are given below. a. Use these data and the ClausiusClapeyron equation (Equation 6.1-3) to estimate the heat of vaporization of chlorine (kJ/mol) and to obtain an...
-
How would you prepare benzyl amine, C6H5CH2NH2, from benzene? More than one step is needed.
-
Peachtree Construction, Inc., which is incorporated as a corporation, reported a NOL of $500,000 in 2008. Given the following data, how much of this NOL is likely to be available as a deduction from...
-
Indo Corporation was organized on January 4 2018, and began active business on January 5 , 2018. Indo incurred the following expenses in connection with creating its business. What is the maximum...
-
Assume Gillette Corporation will pay an annual dividend of $0.66 one year from now. Analysts expect this dividend to grow at 11.9% per year thereafter until the fifth year. After then, growth will...
-
When must a dummy variable be used in a model? Provide your own original example of a dummy variable that might be used in a regression model.
-
ABC company is interested in forecasting quarterly sales for year 2017 for one of its products. Based on past 3 years of quarterly sales data (2014, 2015, and 2016), two forecasting methods look...
-
Valley Company's adjusted trial balance on August 31, 2017, its fiscal year-end, follows. On August 31, 2016, merchandise inventory was $25,400. Supplementary records of merchandising activities for...
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
A full journal bearing is 25 mm long. The shaft journal has a diameter of 50 mm with a unilateral tolerance of 0.01 mm. The bushing bore has a diameter of 50.05 mm with a unilateral tolerance of 0.01...
-
A 1 - 1 - in sleeve bearing supports a load of 700 lbf and has a journal speed of 3600 rev/min. An SAE 10 oil is used having an average temperature of 160F. Using Fig. 1216, estimate the radial...
-
A full journal bearing has a shaft diameter of 80.00 mm with a unilateral tolerance of 0.01 mm. The l/d ratio is unity. The bushing has a bore diameter of 80.08 mm with a unilateral tolerance of 0.03...
-
a) Solve cos x = 2xy and cos xy = 2x to 5 decimal places with an initial guess of x0 = 0.5 and yo= 0.5 using proper method. (90 Point) ATTENTION: Please add a comment line to each line of code...
-
Write a program that will asks the user to input 15 students test score then store them in an array named "Score" your program should accomplish the followings: 1. Calculate and display the average....
-
4. What is clock synchronization? Synchronize the network given below when the server. advances 10 second using Berkeley algorithm. Server 2:35 2:00 2:20 1:55 Client 1 Client 2 Client 3

Study smarter with the SolutionInn App


