The surface tension of a liquid is being measured with a ring as shown. The ring has
Question:
The surface tension of a liquid is being measured with a ring as shown. The ring has an outside diameter of 10 cm and an inside diameter of 9.5 cm. The mass of the ring is 10 g. The force required to pull the ring from the liquid is the weight corresponding to a mass of 16 g. What is the surface tension of the liquid (in N/m)?
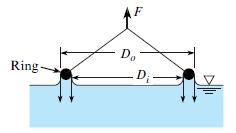
D. Ring- D -
Step by Step Answer:
Mean radius 1095 2 975 cm Net ...View the full answer



Engineering Fluid Mechanics
ISBN: 9781118880685
11th Edition
Authors: Donald F. Elger, Barbara A. LeBret, Clayton T. Crowe, John A. Robertson
Related Video
The property of the surface of a liquid that allows it to resist an external force, due to the cohesive nature of its molecules. The cohesive forces between liquid molecules are responsible for the phenomenon known as surface tension. The molecules at the surface of a glass of water do not have other water molecules on all sides of them and consequently, they cohere more strongly to those directly associated with them (in this case, next to and below them, but not above). is not really true that a \\\"skin\\\" forms on the water surface; the stronger cohesion between the water molecules as opposed to the attraction of the water molecules to the air makes it more difficult to move an object through the surface than to move it when it is completely submersed
Students also viewed these Engineering questions
-
The surface tension of a liquid can be determined by measuring the force F needed to just lift a circular platinum ring of radius r from the surface of the liquid. (a) Find a formula for y in terms...
-
A cylindrical pressure vessel has an outside diameter of 10 in and a wall thickness of 3/8 in. If the internal pressure is 350 psi, what is the maximum shear stress in the vessel walls?
-
A carbon steel tube has an outside diameter of 1 in and a wall thickness of 1/8 in. The tube is to carry an internal hydraulic pressure given as p = N (6000, 500) psi. The material of the tube has a...
-
In a new margin account, an investor sells short 1,000 shares of XYZ stock at $80 per share. If the market value of the stock falls to $70, if the leverage ratio is 1.53846 1. what is the initial...
-
Find the cost of ending inventory using the following table showing Cost per unt Units Total cost $21 $27 $28 $21 13 64 29 48
-
Which of the following are electron-transfer reactions? For those that are, indicate which reactant is the reducing agent and which reactant is the oxidizing agent. (a) 3H 2 SO 3 + 2HNO 3 3H 2 SO 4...
-
Gravity-powered roller coasters have a motorized chain assembly that hauls the cars up to the top of the first hill. No additional source energy is supplied for the rest of the trip. What is the...
-
Solve the requirements of Problem 11.5 for the year ended December 27, 2008. Required: Prepare a common size balance sheet at December 29, 2007, using the following captions: Total current assets...
-
7. Draw the Gantt chart and calculate average waiting time for the following processes using round robin scheduling with time quantum-4. [The information of the processes are given in the image. Draw...
-
The accompanying data for the S&P 500 provides open, high, low, and close values for the S&P index over a period of time. Complete parts a through c below. Click the icon to view the S&P Index. a....
-
A drop of water at 20C is forming under a solid surface. The configuration just before separating and falling as a drop is shown in the figure. Assume the forming drop has the volume of a hemisphere....
-
The local atmospheric pressure is 91 kPa. A gage on an oxygen tank reads a pressure of 250 kPa gage. What is the pressure in the tank in kPa abs?
-
The net income of Novis Corporation is $85,000. The company has 25,000 outstanding shares and a 100 percent payout policy. The expected value of the firm one year from now is $1,725,000. The...
-
In the industrial industry, do you think it's important for them to implement life cycle cost analysis? Explain
-
In 1879, Tesla applied the idea of using high voltages during transmission over distance and lower voltages for home and office using an electromagnetic transformer. a. What would the ratio of turns...
-
Rainbow Company is using both debt and equity financing. Its target capital structure is to achieve 30 percent debt and 70% equity. Early this year, the company invested in project A that provided an...
-
Determine the amount of money earned in an interest bearing account at 4% over 5 years and compounded monthly, if the principal was $20,000
-
How many years will it take $10,000 to grow into one million dollars if it is invested at 4.35% compounded semi-annually?
-
In what order would the following amino acids be eluted with a buffer of pH 4 from a column containing an anion-exchange resin? histidine, serine, aspartate, valine
-
How do the principles of (a) Physical controls and (b) Documentation controls apply to cash disbursements?
-
A 20 m long bored concrete pile having a diameter of 350 mm is load tested, and the data are given below: Also given: E concrete = 25 10 3 MN/m 2 . a. Estimate the allowable pile load using...
-
A steel pile (H-section; HP 310 125; see Table 12.1a) is driven into a layer of sandstone. The length of the pile is 25 m. Following are the properties of the sandstone: unconfined compression...
-
A 600 mm diameter and 25 m long driven concrete pile carries a column load of 1200 kN. It is estimated that the shaft carries 900 kN and the point carries 300 kN. Determine the settlement of the pile...
-
On the bunny hill at a ski resort, a towrope pulls the skiers up the hill with constant speed of 1.87 m/s. The slope of the hill is 10.9 with respect to the horizontal. A child is being pulled up the...
-
A 2.00-mol sample of hydrogen gas is heated at constant pressure from 290 K to 412 K. (a) Calculate the energy transferred to the gas by heat. 4.862 You can use the tabulated value of the specific...
-
Compute for the adjusted balances of the following: Cash on hand Petty Cash Fund BPI Current Account Cash and Cash Equivalents You were able to gather the following from the December 31, 2020 trial...

Study smarter with the SolutionInn App


