(a) Use Equation 35.8 to draw an energy-level diagram for the first six energy levels of a...
Question:
(a) Use Equation 35.8 to draw an energy-level diagram for the first six energy levels of a particle in a cubical box, in terms of h2 /8mL2 ,?(b) give the degeneracy of each.
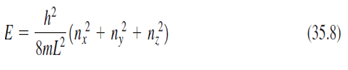
Transcribed Image Text:
E 8ml? (m? + n} + a?) (35.8)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
That means the particle is bounded in some region Then if we get a zero energy state means energy ...View the full answer

Answered By

Hardik Dudhat
I am semi-qualified Chemical Engineering ,I have scored centum in accounting in my senior secondary and in my graduation. I have always helped my fellow students with their concerns on the subject, i have tutored on various tutoring sites in the past and also have taken home tuitions for degree and MBA students. As a tutor, I don't want my students to just get a solution, I want them to understand the concept and never have a doubt in that area thereon and i believe in excelling and not in educating.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw an energy level diagram for the excited state of H2. Is there still a bond between the hydrogens?
-
Use your result from P15.17 and make an energy level diagram for the first five energy levels of a square two dimensional box of edge length b. Indicate which of the energy levels are degenerate and...
-
Draw an energy level diagram for the chemical reaction in Exercise 9. In exercise 9, Nitrogen gas reacts with oxygen gas to make NO(g) while absorbing 180 kJ. Is this process exothermic or...
-
Suri recently took a job in the marketing department for All Access Cable Company. After just a few days on the job. After just a few days on the job, Suri learned that the company has many layers of...
-
A chemist, Ilov Boronin, carried out a reaction of frans-2-pentene with BH3 in THF followed by treatment with H2O2/-OH. Two products were separated and isolated. Desperate to know their structures,...
-
Heteroskedastic individual effects. (a) For the one-way error component model with heteroskedastic \(\mu_{i}\), i.e., \(\mu_{i} \sim\left(0, w_{i}^{2}ight)\), verify that \(\Omega=E\left(u...
-
North Shore Architectural Stone, Inc., a company that installs limestone in residential and commercial buildings, agreed to supply and install limestone for a property owned by Joseph Vitacco. North...
-
Marston Marble Corporation is considering a merger with the Conroy Concrete Company. Conroy is a publicly traded company, and its beta is 1.30. Conroy has been barely profitable, so it has paid an...
-
A spherical beach pebble is completely submerged under sea water of mass density (not weight density!) 1041 kg/m3. The water exerts an upward buoyant force of magnitude 181 N on the pebble. What is...
-
Mike Greenberg opened Kleene Window Washing Inc. on July 1, 2022. During July, the following transactions were completed. July 1 Issued 12,000 shares of common stock for $12,000 cash. 1 Purchased...
-
A particle of mass m is in a region where its total energy E is less than its potential energy U. Show that the Schr?dinger equation has nonzero solutions of the form Ae? .?Such solutions describe...
-
The generalization of the Schrdinger equation to three dimensions is For a particle confined to the cubical region 0 x L, 0 y L, 0 z L, show by direct substitution that the equation is...
-
In Figure, assume that the cylinder rolls without slipping. The spring is at its free length when x and y are zero, (a) Derive the equation of motion in terms of x, with y(t) as the input, (b)...
-
4. For the given relational schema identify the FDs. Based on the FDs identify the PK. Cust id Cust name Cust addr Acc type balance Accno ACC100 C100 John Vellore Savings 100000 C200 Peter Chennai...
-
a. You purchase a stock for $9,000 and collect $300 at the end of each year in dividends. You sell the stock for $10,500 after five years. What was the annual return on your $9,000 investment? %= b....
-
Last year, Rebecca's Bookstore had $1 million in sales. She spent $590,000 to buy the books that she sold and paid out $300,000 in operating expenses. Interest and taxes came to $3,000. What was...
-
3. What Kind of Bear are You? [25 points] You are at your cabin in the deep woods and have taken many pictures of bears - some of them are grizzly bears, some of them are brown bears, and some of...
-
What are the possible consequences of accepting inaccurate methodology of computing cost of capital on AES? Explain
-
What is the distinguishing feature of an excise tax?
-
Refrigerant-134a enters an adiabatic compressor as saturated vapor at 120 kPa at a rate of 0.3 m3/min and exits at 1-MPa pressure. If the isentropic efficiency of the compressor is 80 percent,...
-
For each amino acid, draw the structure that predominates at the isoelectric point: (a) l-Glutamine (b) l-Phenylalanine (c) l-Proline (d) l-Threonine
-
Optically active amino acids undergo racemization at the position when treated with strongly basic conditions. Provide a mechanism that supports this observation.
-
A mixture containing l-glycine, l-glutamine, and l-asparagine was subjected to electrophoresis. Identify which of the amino acids moved the farthest distance assuming that the experiment was...
-
Gilbert Canned Produce (GCP) packs and sells three varieties of canned produce: green beans; sweet peas; and tomatoes. The company is currently operating at 82 percent of capacity. Worried about the...
-
The accounting records of Allen Insulation, Inc. reflected the following balances as of January 1, 20x0: Beginning Inventory: $190 units @ $120 The following transactions occurred in 20x0: January...
-
Sarasota Limited has a calendar-year accounting period. The following errors were discovered in 2023. 1. The December 31, 2021 merchandise inventory had been understated by $50,400. 2. 3. Merchandise...

Study smarter with the SolutionInn App


