The accompanying data were obtained in a slope-ratio investigation of the complex formed between Ni 2+ and
Question:
The accompanying data were obtained in a slope-ratio investigation of the complex formed between Ni2+ and 1-cyclopentene-1-dithiocarboxylic acid (CDA). The measurements were made at 530 nm in 1.00-cm cells.
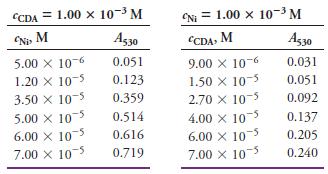
(a) Determine the formula of the complex. Use linear least squares to analyze the data.
(b) Find the molar absorptivity of the complex and its uncertainty.
Transcribed Image Text:
CCDA = 1.00 x 10-3 M (NI = 1.00 x 10-3 M Ni M Aş3o CDA, M Ag30 5.00 x 10-6 1.20 X 10-5 3.50 X 10-5 0.051 9.00 X 10-6 1.50 X 10-5 2.70 X 10-5 4.00 X 105 0.031 0.123 0.051 0.359 0.092 5.00 X 10-5 6.00 X 10 7.00 x 105 0.514 0.137 0.616 6.00 X 105 7.00 X 105 0.205 0.719 0.240
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 36% (11 reviews)

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The following data were obtained in a BET apparatus for adsorption equilibrium of nitrogen on silica gel (SG) at -195.8C. Estimate the specific surface area in m2/g of silica gel. How does your value...
-
In a recent survey, the following data were obtained in response to the question, If the number of summer classes were increased, would you be more likely to enroll in one or more of them? If a...
-
The following data were obtained in a study of the relationship between the weight and chest size of infants at birth: Weight (kg) Chest Size (cm) 275...................................29.5...
-
Implement a generic Map that supports the put and get operations. The implementation will store a hash table of pairs (key, definition). Figure 5.55 provides the Map specification (minus some...
-
This problem demonstrates the dependence of the future value of an annuity on the number of payments. Suppose $1000 is invested at the end of each year. Assume the investments earn 10% compounded...
-
Slow n Steady, Inc., has a stock price of $34, will pay a dividend next year of $3.25, and has expected dividend growth of 1.3% per year. What is your estimate of Slow n Steadys cost of equity...
-
The pressure distribution measured along a straight, horizontal portion of a 50-mm-diameter pipe attached to a tank is shown in the table below. Approximately how long is the entrance length? In the...
-
The Morning Brew Coffee Shop sells Regular, Cappuccino, and Vienna blends of coffee. The shop's current daily labor cost is $320, the equipment cost is $125, and the overhead cost is $225. Daily...
-
An isotropic point source emits S neutrons/s in an infinite moderator (i.e., low absorbing and highly scattering). PART 3A): Obtain the formulation, which upon solution yields the total number of...
-
Changs Office Furniture manufactures office furniture by using an assembly-line process. All direct materials are introduced at the start of the process, and conversion cost is incurred evenly...
-
The accompanying absorption data were recorded at 390 nm in 1.00-cm cells for a continuous-variations study of the colored product formed between Cd 2+ and the complexing reagent R. (a) Find the...
-
The sodium salt of 2-quinizarinsulfonic acid (NaQ) forms a complex with Al 3+ that absorbs strongly at 560 nm.19 The data collected on this system are shown in the accompanying table. (a) Find the...
-
What were the major factors that led to the drop in Sony's exports from Japan?
-
Consider the beam shown in the diagram below: 5 N 30 1 m 1m A 2 m 15 N 10 N 3m 3 N/m B Am TTT 2 m List the support type at Points A and B. b. Determine the equivalent point load magnitude of the...
-
Describe the relationship between the governmental agencies (SEC and Federal Reserve) and the banks (Bank of America, JP Morgan, Merrill Lynch, Wachovia etc. How did they view their responsibilities?...
-
Why is it better to notify someone in person when they are terminated? It allows the manager to address shortcomings. It ensures consistency. It meets legal requirements. It shows respect for the...
-
Mike contributed $50,000 in exchange for a general partnership interest and Ned contributed $50,000 in exchange for a limited partnership interest. The partnership agreement provides that Mike and...
-
Borden Car Sales had sales of $125,023 in March and $112,321 in April. Calculate the rate of change in sales from March to April.
-
A new drug is being compared with a standard drug for treating a particular illness. In the clinical trials, a group of 200 patients was randomly split into two groups, with one group being given the...
-
Rowland Textile Inc. manufactures two products: sweatshirts and T-shirts. The manufacturing process involves two activities: cutting and sewing. Expected overhead costs and cost drivers are as...
-
Calculate the equivalent mass of oxalic acid dehydrate (H2C2O4 2H2O, 126.066 g/mol) when it is titrated to (a) A bromocresol green end point and (b) A phenolphthalein end point.
-
Give two reasons why KH(IO3)2 is preferred over benzoic acid as a primary standard for a 0.010 M NaOH solution.
-
Briefly describe the circumstance where the concentration of a sodium hydroxide solution will apparently be unaffected by the absorption of carbon dioxide.
-
Design a power transmission system to connect a DC motor to its load. The motor is given in the motor catalogue as a BM1400 motor. You must create a transmission which will connect to this motor and...
-
SDJ, Incorporated, has net working capital of $3,320, current liabilities of $4,550, and inventory of $4,600. a. What is the current ratio? b. What is the quick ratio?
-
Calculate the final selling price to the nearest cent and markdown percent to the nearest hundredth percent: Original selling price $4,900 First markdown 20% Second markdown 10% Markup 12% Final...

Study smarter with the SolutionInn App


