The following data were obtained in a BET apparatus for adsorption equilibrium of nitrogen on silica gel
Question:
The following data were obtained in a BET apparatus for adsorption equilibrium of nitrogen on silica gel (SG) at -195.8°C. Estimate the specific surface area in m2/g of silica gel. How does your value compare with that in Table15.2?
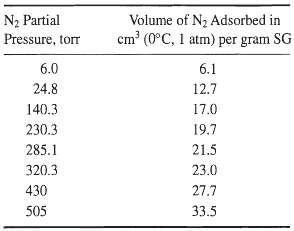
Transcribed Image Text:
N2 Partial Pressure, torr Volume of N2 Adsorbed in cm (0°C, 1 atm) per gram SG 6.0 6.1 24.8 12.7 140.3 17.0 19.7 230.3 285.1 21.5 23.0 320.3 430 27.7 33.5 505
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
From Eq 156 where P total pressure partial pressure of N 2 in the ab...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
12a. Given the following SEL-351 circuit and the following description, provide settings for the relay: The breaker should trip if the current rises above 5.7 amps secondary if the breaker is closed...
-
In a recent survey, the following data were obtained in response to the question, If the number of summer classes were increased, would you be more likely to enroll in one or more of them? If a...
-
The following data were obtained from an independentmeasures research study comparing three treatment conditions. Use an ANOVA with a = .05 to determine whether there are any significant mean...
-
Determine the order of growth of the running time of this statement in ThreeSum as a function of the number of integers \(n\) on standard input: int[] a = StdIn. readA11Ints(); Answer: Linear. The...
-
Glade, Marker, and Walters are partners with beginning-year capital balances of $100,000, $50,000, and $50,000, respectively. Partnership net income for the year is $84,000. Make the necessary...
-
Given the translational mechanical system of Figure P4.20, where K = 1 and f(t) is a unit step, find the values of M and f v to yield a response with 17% overshoot and a settling time of 10 seconds....
-
Consider the automobile gasoline mileage data in Table B.3. a. Build a linear regression model relating gasoline mileage \(y\) to engine displacement \(x_{1}\) and the type of transmission...
-
Your retirement fund consists of a $5,000 investment in each of 15 different common stocks. The portfolio's beta is 1.20. Suppose you sell one of the stocks with a beta of 0.8 for $5,000 and use the...
-
3) (10 pts) Write a function NextMinute that receives one integer as the input argument, which is the current minute of the hour, and returns the next minute as the output argument. Please note that...
-
On January 1, 2020, Pinnacle Corporation exchanged $3.561,500 cash for 100 percent of the outstanding voting stock of Strata Corporation. On the acquisition date, Strata had the following balance...
-
Representative properties of small-pore silica gel are as follows: pore diameter = 24 A; particle porosity = 0.47; particle density = 1.09 g/cm3 and specific surface area = 800 m2/g (a) Are these...
-
Estimate the maximum ion-exchange capacity in meq/g resin for an ion-exchange resin made from 8 wt% divinylbenzene and 92 wt% styrene.
-
Water at 1.5 MPa, 150C, is throttled adiabatically through a valve to 200 kPa. The inlet velocity is 5 m/s, and the inlet and exit pipe diameters are the same. Determine the state and the...
-
What is the result of executing the HopCounter program? A. The code prints hop once. B. The code prints hop twice. C. The first compiler error is on line p1. D. The first compiler error is on line...
-
Which statements about abstract classes and methods are correct? (Choose three.) A. An abstract class can be extended by a final class. B. An abstract method can be overridden by a final method. C....
-
Which of the following is a valid method name in Java? (Choose two.) A. Go_$Outside$2() B. have-Fun() C. new() D. 9enjoyTheWeather() E. $sprint() F. walk#()
-
Which statements about Java classes are true? (Choose three.) A. A Java class file may include more than one package statement. B. A Java class file may include more than one import statement. C. A...
-
What is the result of executing the Tortoise program? A. init-hare-tortoise B. init-hare C. The first line with a compiler error is line x1. D. The first line with a compiler error is line x2. E. The...
-
Why are pens used in applications?
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
Locate the Treasury bond in Figure 8.5 maturing in November 2041. Is this a premium or a discount bond? What is its current yield? What is its yield to maturity? What is the bid-ask spread in...
-
Liquid isobulune is throttled through a valve from an initial state of 360 K and 4,000 kPla to a final pressure of 2,000 kPa. Estimate the temperature change and the entropv change of the isobutane....
-
A stream of air at 12 bar and 900 K is mixed with another stream of air at 2 bar and 400 K with 2.5 times the mass flowrate, If this process were accomplished reversibly and adiabatically, what would...
-
Hot nitrogen gas at 750(oF) and atmospheric pressure flows into a waste-heat boiler at the rate of 40 (Ibm) (s)-1, and transfers heat to water boiling at l(atm). The water feed to the boiler Is...
-
Hinrich Company traded machinery with a book value of $120,000 and a fair value of $200,000. It received in exchange from Noach Company a machine with a fair value of $180,000 and cash of $20,000....
-
You have just moved from Norfolk, Virginia (sea level), to Taos, New Mexico (high in the mountains), and you find yourself out of breath climbing a small hill. Three months later, climbing the same...
-
Starline is a small children's clothing manufacturer and retailer that has seen rapid growth in the last twelve months and a significant increase in employees. At a meeting of managers, a number of...

Study smarter with the SolutionInn App


