A gas has C P * = 35 J/mol K, and follows the equation of state:
Question:
A gas has CP* = 35 J/mol · K, and follows the equation of state: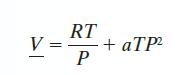 with a = 0.15 cm3/mol · bar2 · K. Find the change in A when the gas is compressed isothermally from T = 300 K and P = 1 bar to P = 10 bar.
with a = 0.15 cm3/mol · bar2 · K. Find the change in A when the gas is compressed isothermally from T = 300 K and P = 1 bar to P = 10 bar.
Transcribed Image Text:
V= RT P + aTP²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
The change in Helmholtz free energy AA for an isotherm...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
An economy is initially described by the following equations C = 500 +0.50(Y-T) 1 = 500-25r d =Y - 25r G = 100 T = 500 M = 2000 P = 4 a. Write down the equations for the IS and LM curves and...
-
A gas has an ideal gas heat capacity of C P * = (7/2)R and is described by the equation of state: Z = 1 + (CP 2 )/(RT) with C = 100 cm 3 /bar mol. A . Find a general expression for the residual...
-
A gas with a flow rate of 300 mol/min enters a steady-state, adiabatic nozzle with negligible velocity at T = 500 K and P = 10 bar and leaves the nozzle at P = 1 bar. The gas has C P * = 40 J/mol K....
-
Name: 9. 11. Bell: Directions: Evaluate each expression using the order of operations. 1. 17-5-4+2 2. 40-32+8+5--2 Date: 5. 2018-(5+3)+7] 3.35-14+2+8 7.1+(-2-5)+(14-17)-4 25 +8+3 4+3 Unit 1: Algebra...
-
Assume you are a member of an international policy setting committee and are responsible for harmonizing audit report requirements internationally. Examine Exhibit 9-8. Based on the varying...
-
What type of inventory are we trying to minimize as the result of workcenter scheduling?
-
Find the solution of a spring-mass-damper system governed by the equation \(m \ddot{x}+c \dot{x}+k x=F(t)=\delta F . t\) with \(m=c=k=1\) and \(\delta F=1\). Assume the initial values of \(x\) and...
-
Cordell Inc. experienced the following events in 2016, its first year of operation: 1. Received $40,000 cash from the issue of common stock. 2. Performed services on account for $82,000. 3. Paid a...
-
Describe the role of the kernel in managing networking operations. How does the kernel handle packet routing, filtering, and network protocol stack operations, and what mechanisms are used to ensure...
-
Imagine a compound has T C = 500 K and P C = 20 bar. Use the Peng-Robinson equation throughout this problem. A. Plot P- V at T = 400 K, T = 500 K, and T = 600 K, assuming the compound has = 0. B....
-
Steam is heated from an initial condition of saturated steam at P = 1.5 bar to a final state of P = 3 bar and T = 3008C. Use the steam tables to find the change in for this process.
-
There is money to send two of eight city council members to a conference in Honolulu. All want to go, so they decide to choose the members to go to the conference by a random process. How many...
-
\(\quad \Lambda(r, \zeta)\) approaches 0 for large \(r\) for all values of \(\zeta\). Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to...
-
The phase angle depends upon \(F_{0}\), which is the amplitude of excitation. Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to make it...
-
An increase in damping leads to an increase in the percentage of isolation. Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to make it...
-
A \(40 \mathrm{~kg}\) mass hangs from a spring with a stiffness of \(4 \times 10^{4} \mathrm{~N} / \mathrm{m}\). A harmonic force with a magnitude of \(120 \mathrm{rad} / \mathrm{s}\) is applied....
-
If \(\phi\) is positive in the equation \(x(t)=X \sin (\omega t-\phi)\), the response lags the excitation. Indicate whether the statement presented is true or false. If true, state why. If false,...
-
FX Corporation provides special effects for movies filmed in Manitoba. The company needs a sophisticated piece of equipment that can only be sourced from the United States. FX submitted a purchase...
-
Find i 0 (t) for t > 0 in the circuit in Fig. 16.72 . 2 + Vo 1 7.5e-2t u(t) V ( +) 4.5[1 u(t)]V 0.5v. 1H
-
Use MATLAB to solve the following problem: x - 3y = 2 x + 5y = 18 4x - 6y = 10
-
a. Use MATLAB to find the coefficients of the quadratic polynomial y = ax 2 + bx + c that passes through the three points (x, y) = (1, 4), (4, 73), (5, 120). b. Use MATLAB to find the coefficients of...
-
Use the MATLAB program given in Table 8.52 to solve the following problems: a. Problem 3d d. b. Problem 11 Solve the following equations: c. Problem 14 Use MATLAB to solve the following problem: x -...
-
2. Construct the cobweb diagram for, a. Nt+1 = (1+r) Nt 1+rNt Nt > 0 b. xn+1 = xn x + 1/1/x/1 Determine the stability of fixed points in each case for any real-valued r. For part (b), find all of the...
-
The following is a transcription of a real conversation with Chat GPT (the artificial intelligence language model that has been in the news lately) that Jerrod had on January 25, 2023. User input:...
-
Nomsa was appointed six months ago as a Secretary in the Department of Finance. During her onboarding meeting she was told she will be a minuting secretary to the Departmental Committee meeting that...

Study smarter with the SolutionInn App


