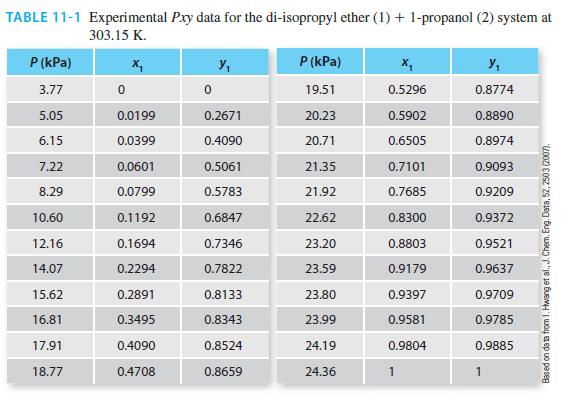
Perform a reduction of the data in Table 11-1 for the di-isopropyl ether (1) + 1-propanol (2)
Question:
Perform a reduction of the data in Table 11-1 for the di-isopropyl ether (1) + 1-propanol (2) system at 303.15 K using both the 1-parameter and 2-parameter Margules equations.
Transcribed Image Text:
TABLE 11-1 Experimental Pxy data for the di-isopropyl ether (1) + 1-propanol (2) system at 303.15 K. X₂ P (kPa) 3.77 5.05 6.15 7.22 8.29 10.60 12.16 14.07 15.62 16.81 17.91 18.77 0 0.0199 0.0399 0.0601 0.0799 0.1192 0.1694 0.2294 0.2891 0.3495 0.4090 0.4708 0 Y₁ 0.2671 0.4090 0.5061 0.5783 0.6847 0.7346 0.7822 0.8133 0.8343 0.8524 0.8659 P (kPa) 19.51 20.23 20.71 21.35 21.92 22.62 23.20 23.59 23.80 23.99 24.19 24.36 0.5296 0.5902 0.6505 0.7101 0.7685 0.8300 0.8803 0.9179 0.9397 0.9581 0.9804 y₁ 0.8774 0.8890 0.8974 0.9093 0.9209 0.9372 0.9521 0.9637 0.9709 0.9785 0.9885 1 Based on data from 1. Hwang et al., J. Chem. Eng. Data, 52, 2503 (2007).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Step 1 Obtain the experimental activity coefficients and the experimental excess molar Gibbs free energy We can obtain the experimental activity coefficients and the excess molar Gibbs free energy in ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
The article Seismic Hazard in Greece Based on Different Strong Ground Motion Parameters (S. Koutrakis, G. Karakaisis, et al., Journal of Earthquake Engineering, 2002:75109) presents a study of...
-
Using the data in Table 116 on page 300, indicate the semiannual interest payment dates for the Motorola bonds that mature in 2031. (For the item in question, look under Interest Dates.) The two...
-
The data in Table P-13 are closing stock quotations for the DEF Corporation for 150 days. Determine the appropriate ARIMA model, and forecast the stock price five days ahead from forecast origin t =...
-
Assume you have just been hired as a business manager of PizzaPalace, a regional pizza restaurant chain. The companys EBIT was $120 million last year and is not expected to grow. PizzaPalace is in...
-
Why is it important for a project to have flexibility?
-
Explain the main conceptual issue under variable costing and absorption costing regarding the timing for the release of fixed manufacturing overhead as expense.
-
Suppose Apex Electrical Supplys Inventory account showed a balance of \($43,000.\) A physical count showed \($41,800\) of goods on hand. To adjust the inventory account, Apex Electrical Supply, Inc.,...
-
A hydrogen atom in the 5g state is placed in a magnetic field of 0.600 T that is in the z-direction. (a) Into how many levels is this state split by the interaction of the atom's orbital magnetic...
-
Today the spot rate is $1.15 per euro and a U.S. investor buys one million dollars of a European mutual fund at a price of 20 per share. One year later the shares have appreciated to 24. What rate of...
-
Estimate the vapor-liquid equilibrium for the n-pentane (1) + benzene (2) system at 313.15 K using the van Laar equation.
-
Estimate the mole fraction of oxygen in water at 25C and at 75C and 1 atm.
-
Obtain an annual report from a public college or university and answer the following questions: a. Does the institution report as a special-purpose entity engaged in (1) governmental- and...
-
On a standardized test with a normal distribution, the mean is 20 and the standard deviation is 2.6. In which interval would the greatest number of scores occur?
-
What is the best description of unregulated capitalism and the drawbacks involved?
-
Based on the information on this website, how you can explain about Situation Analysis for this product...
-
Kaya, Karatas, Gurcan, and Dalgic's "A grounded theory study to explore the digital gameplay experiences of adolescents at risk of addiction in Turkey" ( Journal of Pediatric Nursing, 63 ,...
-
Assume a project has projected annual depreciation of $878, annual fixed costs of $3,200, and a variable cost per unit of $8.21. The sales price per unit is expected to be $13.39. What is the...
-
Plot the following data, and from the graph determine ÎHvap for magnesium and lithium. In which metal is the bonding stronger? Vapor Pressure mm Hg) Temperature C) Mg Li 10 100 400 760 750 890...
-
DC has unused FTC carryover from 2017 in the separate category for GC income as the result of income generated by a foreign branch. The income was foreign source general category income. In 2018 the...
-
Use MuPAD to compute the following limits. x + 1 lim x- 00 . 3x b. lim 2x 2x + 3 X -00
-
Find the expression for the sum of the geometric series for r 1. n-1 k=0
-
A particular rubber ball rebounds to one-half its original height when dropped on a floor . a. If the ball is initially dropped from a height h and is allowed to continue to bounce, find the...
-
s On January 1, 2024, Adams-Meneke Corporation granted 120 million incentive stock options to division managers, each permitting holders to purchase one share of the company's $1 par common shares...
-
CompuTech, Inc., a manufacturer of products using the latest microprocessor technology, has appointed you the manager of its Micro Technology Division. Your division has $800000 in assets and...
-
Ex. Assume a person uses Priceline.com to bid $100 on a rental car for one week. The average rental rate for one week is $250. Assume that Hertz rental car has excess capacity for that week. The...

Study smarter with the SolutionInn App


