Use the data in the steam tables (Appendix A) to answer the following: A. Find the change
Question:
Use the data in the steam tables (Appendix A) to answer the following:
A. Find the change in internal energy when 50 kg of steam at constant pressure P = 5 bar has its temperature reduced from 500°C to 300°C.
B. Find the change in internal energy when 200 kg of liquid water at constant pressure P = 100 bar has its temperature increased from 240°C to 300°C.
Appendix A.
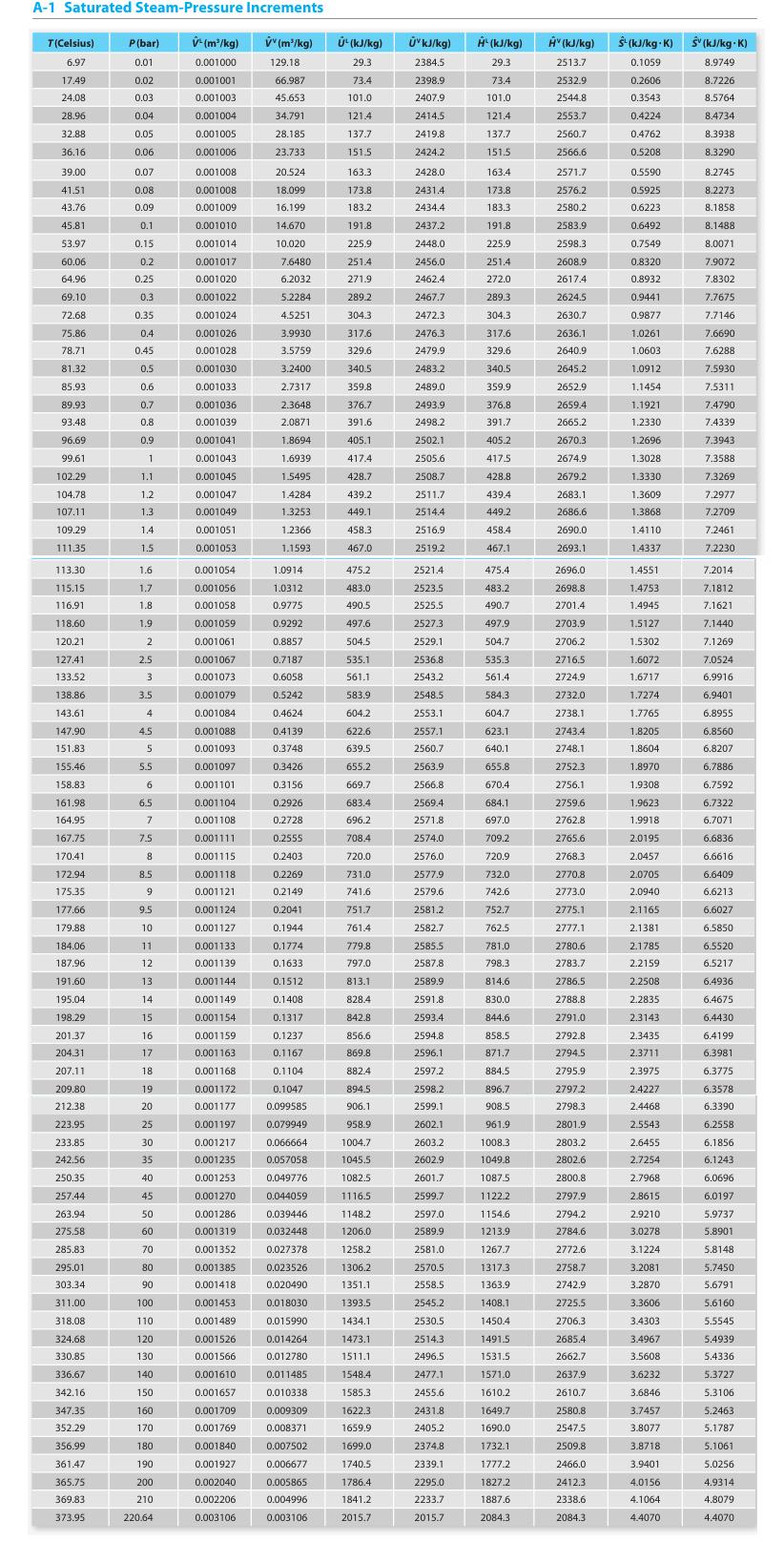
Transcribed Image Text:
A-1 Saturated Steam-Pressure Increments T(Celsius) 6.97 17.49 24.08 28.96 32.88 36.16 39.00 41.51 43.76 45.81 53.97 60.06 64.96 69.10 72.68 75.86 78.71 81.32 85.93 89.93 93.48 96.69 99.61 102.29 104.78 107.11 109.29 111.35 113.30 115.15 116.91 118.60 120.21 127.41 133.52 138.86 143.61 147.90 151.83 155.46 158.83 161.98 164.95 167.75 170.41 172.94 175.35 177.66 179.88 184.06 187.96 191.60 195.04 198.29 201.37 204.31 207.11 209.80 212.38 223.95 233.85 242.56 250.35 257.44 263.94 275,58 285.83 295.01 303.34 311.00 318.08 324.68 330.85 336.67 342.16 347.35 352.29 356.99 361.47 365.75 369.83 373.95 P(bar) 0.01 0.02 0.03 0.04 0.05 0.06 0.07 0.08 0.09 0.1 0.15 0.2 0.25 0.3 0.35 0.4 0.45 0.5 0.6 0.7 0.8 0.9 1 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 2 2.5 3 3.5 4 4.5 5 5.5 6 6.5 7 7.5 8 8.5 9 9.5 10 11 12 13 14 15 16 17 18 19 20 25 30 35 40 45 50 60 70 80 90 100 110 120 130 140 150 160 170 180 190 200 210 220.64 V¹ (m³/kg) 0.001000 0.001001 0.001003 0.001004 0.001005 0.001006 0.001008 0.001008 0.001009 0.001010 0.001014 0.001017 0.001020 0.001022 0.001024 0.001026 0.001028 0.001030 0.001033 0.001036 0.001039 0.0010411 0.001043 0.001045 0.001047 0.001049 0.001051 0.001053 0.001054 0.001056 0.001058 0.001059 0.001061 0.001067 0.001073 0.001079 0.001084 0.001088 0.001093 0.001097 0.001101 0.001104 0.001108 0.001111 0.001115 0.001118 0.001121 0.001124 0.001127 0.001133 0.001139 0.001144 0.001149 0.001154 0.001159 0.001163 0.001168 0.001172 0.001177 0.001197 0.001217 0.001235 0.001253 0.001270 0.001286 0.001319 0.001352 0.001385 0.001418 0.001453 0.001489 0.001526 0.001566 0.001610 0.001657 0.001709 0.001769 0.001840 0.001927 0.002040 0.002206 0.003106 vv (m³/kg) 129.18 66.987 45.653 34.791 28.185 23.733 20.524 18.099 16.199 14.670 10.020 7.6480 6.2032 5.2284 4.5251 3.9930 3.5759 3.2400 2.7317 2.3648 2.0871 1.8694 1.6939 1.5495 1.4284 1.3253 1.2366 1.1593 1.0914 1.0312 0.9775 0.9292 0.8857 0.7187 0.6058 0.5242 0.4624 0.41391 0.3748 0.3426 0.3156 0.2926 0.2728 0.2555 0.2403 0.2269 0.2149 0.2041 0.1944 0.1774 0.1633 0.1512 0.1408 0.1317 0.1237 0.1167 0.1104 0.1047 0.099585 0.079949 0.066664 0.057058 0.049776 0.044059 0.039446 0.032448 0.027378 0.023526 0.020490 0.018030 0.015990 0.014264 0.012780 0.011485 0.010338 0.009309 0.008371 0.007502 0.006677 0.005865 0.004996 0.003106 U¹(kJ/kg) 29.3 73.4 101.0 121.4 137.7 151.5 163.3 173.8 183.2 191.8 225.9 251.4 271.91 289.2 304.3 317.6 329.6 340.5 359.8 376.7 391.6 405.1 417.4 428.7 439.2 449.1 458.3 467.0 475.2 483.0 490.5 497.6 504.5 535.1 561.1 583.9 604.2 622.6 639.5 655.2 669.7 683.4 696.2 708.4 720.0 731.0 741.6 751.7 761.4 779.8 797.0 813.1 828.4 842.8 856.6 869.8 882.4 894.5 906.1 958.9 1004.7 1045.5 1082.5 1116.5 1148.2 1206.0 1258.2 1306.2 1351.1 1393.5 1434.1 1473.1 1511.1 1548.4 1585.3 1622.3 1659.9 1699.0 1740.5 1786.4 1841.2 2015.7 Úv kJ/kg) 2384.5 2398.9 2407.9 2414.5 2419.8 2424.2 2428.0 2431.4 2434.4 2437.2 2448.0 2456.0 2462.4 2467.7 2472.3 2476.3 2479.9 2483.2 2489.0 2493.9 2498.2 2502.1 2505.6 2508.7 2511.7 2514.4 2516.9 2519.2 2521.4 2523.5 2525.5 2527.3 2529.1 2536.8 2543.2 2548.5 2553.1 2557.1 2560.7 2563.9 2566.8 2569.4 2571.8 2574.0 2576.0 2577.9 2579.6 2581.2 2582.7 2585.5 2587.8 2589.9 2591.8 2593.4 2594.8 2596.1 2597.2 2598.2 2599.1 2602.1 2603.2 2602.9 2601.7 2599.7 2597.0 2589.9 2581.0 2570.5 2558.5 2545.2 2530.5 2514.3 2496.5 2477.1 2455.6 2431.8 2405.2 2374.8 2339.1 2295.0 2233.7 2015.7 H' (kJ/kg) 29.3 73.4 101.0 121.4 137.7 151.5 163.4 173.8 183.3 191.8 225.9 251.4 272.0 289.3 304.3 317.6 329.6 340.5 359.9 376.8 391.7 405.2 417.5 428.8 439.4 449.2 458.4 467.1 475.4 483.2 490.7 497.9 504.7 535.3 561.4 584.3 604.7 623.1 640.1 655.8 670.4 684.1 697.0 709.2 720.9 732.0 742.6 752.7 762.5 781.0 798.3 814.6 830.0 844.6 858.5 871.7 884.5 896.7 908.5 961.9 1008.3 1049.8. 1087.5 1122.2 1154.6 1213.9 1267.7 13173 1363.9 1408.1 1450.4 1491.5 1531.5 1571.0 1610.2 1649.7 1690.0 1732.1 1777.2 1827.2 1887.6 2084.3 Hv (kJ/kg) 2513.7 2532.9 2544,8 2553.7 2560.7 2566.6 2571.7 2576.2 2580.2 2583.9 2598.3 2608.9 2617.4 2624.5 2630.7 2636.1 2640.9 2645.2 2652.9 2659.4 2665.2 2670.3 2674.9 2679.2 2683.1 2686.6 2690.0 2693.1 2696.0 2698.8 2701.4 2703.9 2706.2 2716.5 2724.9 2732.0 2738.1 2743.4 2748.1 2752.3 2756.1 2759.6 2762.8 2765.6 2768.3 2770.8 2773.0 2775.1 2777.1 2780.6 2783.7 2786.5 2788.8 2791.0 2792.8 2794.5 2795.9 2797.2 2798.3 2801.9 2803.2 2802.6 2800.8 2797.9 2794.2 2784.6 2772.6 2758.7 2742.9 2725.5 2706.3 2685.4 2662.7 2637.9 2610.7 2580.8 2547.5 2509.8 2466.0 2412.3 2338.6 2084.3 S¹ (kJ/kg-K) S (kJ/kg-K) 0.1059 8.9749 0.2606 8.7226 0.3543 8.5764 0.4224 0.4762 0.5208 0.5590 0.5925 0.6223 0.6492 0.7549 0.8320 0.8932 0.9441 0.9877 1.0261 1.0603 1.0912 1.1454 1.1921 1.2330 1.2696 1.3028 1.3330 1.3609 1.3868 1.4110 1.4337 1.4551 1.4753 1.4945 1.5127 1.5302 1.6072 1.6717 1.7274 1.7765 1.8205 1.8604 1.8970 1.9308 1.9623 1.9918 2.0195 2.0457 2.0705 2.0940 2.1165 2.1381 2.1785 2.2159 2.2508 2.2835 2.3143 2.3435 2.3711 2.3975 2.4227 2.4468 2.5543 2.6455 2.7254 2.7968 2.8615 2.9210 3.0278 3.1224 3.2081 3.2870 3.3606 3.4303 3.4967 3.5608 3.6232 3.6846 3.7457 3.8077 3.8718 3.9401 4.0156 4.1064 4.4070 8.4734 8.3938 8.3290 8.2745 8.2273 8.1858 8.1488 8.0071 7.9072 7.8302 7.7675 7.7146 7.6690 7.6288 7.5930 7.5311 7.4790 7.4339 7.3943 7.3588 7.3269 7.2977 7.2709 7.2461 7.2230 7.2014 7.1812 7.1621 7.1440 7.1269 7.0524 6.9916 6.9401 6.8955 6.8560 6.8207 6.7886 6.7592 6.7322 6.7071 6.6836 6.6616 6.6409 6.6213 6.6027 6.5850 6.5520 6.5217 6.4936 6.4675 6.4430 6.4199 6.3981 6.3775 6.3578 6.3390 6.2558 6.1856 6.1243 6.0696 6.0197 5.9737 5.8901 5.8148 5.7450 5.6791 5.6160 5.5545 5.4939 5.4336 5.3727 5.3106 5.2463 5.1787 5.1061 5.0256 4.9314 4.8079 4.4070
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Abigael martinez
I have been a tutor for over 3 years and have had the opportunity to work with students of all ages and backgrounds. I have a strong belief that all students have the ability to learn and succeed if given the right tools and support. I am patient and adaptable, and I take the time to get to know each student's individual learning style in order to best support their needs. I am confident in my ability to help students improve their grades and reach their academic goals.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Use the data in the steam tables to answer the following: A . Find the change in internal energy when 100 kg of steam at constant pressure P = 1 bar has its temperature reduced from 300C to 100C. B ....
-
Use the data in the steam tables to answer the following: A. Find the change in volume when 50 kg of steam, initially at T = 200C and P = 2 bar, is heated and compressed into steam at T = 350C and P...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Describe the risk assessment approach used for Enterprise Risk Management(ERM) at Worker's Compensation Fund(WCF). How does this approach compare to ISO 31000? Does it consider risks with upside...
-
How can variable costing reports prepared using the contribution margin format help managers in computing breakeven volume in units?
-
What are the components of a single nucleosome? a. About 146 bp of DNA and four core histone proteins b. About 146 bp of DNA and eight core histone proteins c. About 200 bp of DNA and four core...
-
Many single women and married couples use donated sperm to conceive children each year. Pennsylvania resident Donna Donovan decided to use donated sperm from Idant Laboratories, a New York sperm bank...
-
Naylor Company had $210,000 of net income in 2013 when the selling price per unit was $150, the variable costs per unit were $90, and the fixed costs were $570,000. Management expects per unit data...
-
8) What does the following method do? Rewrite it so it produces the same results but does not use recursion. public static boolean whoKnows (int arr, int i, int j) { if (i >= j) { return true; } else...
-
Jackie serves as the vice president for network development for a large, midwestern healthcare system. She has worked with many rural and semirural hospitals to improve efficiency by offering shared...
-
Your team is designing a process that has six streams containing liquid water or steam at different temperatures and pressures, and you need to know the specific internal energy for each. Your...
-
On a stove, water is heated from room temperature T = 25C to boiling temperature T = 100C. Determine T in degrees Celsius and in degrees Kelvin.
-
After the accounts have been adjusted at October 31, the end of the fiscal year, the following balances were taken from the ledger of Smart Delivery Services Co.: Journalize the four entries required...
-
Why does the matter of representation matter so much for Indigenous peoples? For example, April exclaims when she reads her adoption document that officials describe the kids as they would animals....
-
A 204 ohms and a 2902 ohms resistor are connected in series with a 10-V battery. What is the voltage across the second resistor?
-
Explain what the code segment below is doing, in general. What is the output of the code? (show the entire, exact output.) for (int i=1; i
-
A manufacturer of jeans is interested in developing an advertisement campaign that will reach four different groups. Advertisement campaigns can be conducted through TV, radio and magazines. The...
-
Your mom is expected to get $140,000 in 15 years when she retires from her job. She, however, wanted to open a shop now to generate a second income when she becomes jobless. In order to finance her...
-
Nature Cosmetics Company applies overhead costs on the basis of machine hours. The overhead rate is computed by analyzing data from the previous year to determine the percentage change in costs....
-
On April 29, 2015, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon has assets with a fair market value of $1.4 million (basis...
-
The soil profile at a site consists of 2 m of sand at the ground level, underlain by 6 m of clay, followed by a very stiff clay stratum that can be assumed to be impervious and incompressible. The...
-
The soil profile at a site consists of a 2.0 m thick sand layer at the top, underlain by a 3.0 m thick clay layer. The water table lies at a depth of 1.0 m below the ground level. The bulk and...
-
A direct shear test is conducted on a 60 mm x 60 mm overconsolidated clay specimen. The loading was very slow, ensuring that there is no pore water pressure development within the specimen (i.e.,...
-
2. The John Amos power plant and the Mitchell power plant are both coal fired and emit SO2, regulators are now trying to determine how to cost-effectively allocate abatement requirements across the...
-
Hawaiian Electric Company (HECO) is a monopoly. Let us suppose that increasing returns to scale (IRTS) exist, such that the provision of electricity a natural monopoly. The relevant curves for...
-
Complete a research summary related to the audit process and the meaning of audit risk. Distinguish between auditing standards and and audit procedures. How is audit risk managed through the...

Study smarter with the SolutionInn App


