Derive the following general transport equation, similar to (2.13), for the case when the quantity depends
Question:
Derive the following general transport equation, similar to (2.13), for the case when the quantity χ depends on r, v, and t,
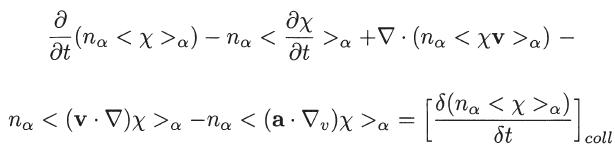
Equation 2.13
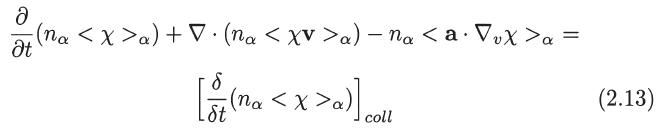
Transcribed Image Text:
Ə
əx
:(na
Ə
əx
:(na a) - na <
Ət
Ət
+V. (na a) -
na < (v⋅V)x >a −na < (a · √₂)X >a
=
5(no a) | coll
[8(na
[8(na
St
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)

Answered By

Daniel Kimutai
I am a competent academic expert who delivers excellent writing content from various subjects that pertain to academics. It includes Electronics engineering, History, Economics, Government, Management, IT, Religion, English, Psychology, Sociology, among others. By using Grammarly and Turnitin tools, I make sure that the writing content is original and delivered in time. For seven years, I have worked as a freelance writer, and many scholars have achieved their career dreams through my assistance.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the general transport equation of the previous problem and let the property (r, v, t) be the random flux of thermal kinetic energy, that is, Show that (considering the Lorentz force for F)...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Mention 4 characteristics of the enzyme systems that are responsible for the biotransformation of xenobiotics.
-
Write a program that takes three double command-line arguments x, y, and z, reads from standard input a sequence of point coordinates (xi, yi, zi), and prints the coordinates of the point closest to...
-
Review the opening feature of this chapter dealing with ash&dans and the entrepreneurial owners, Ashley Cook and Danielle Dankner. Required 1. Assume that ash&dans sells a $300 gift certificate to a...
-
Kate Stephens, the COO of BioDerm, has asked her cost management team for a product-line profitability analysis for her company's two products, Xderm and Yderm. The two skin-care products require a...
-
If the pion slows down to \(0.99990 c\), about what percentage of its kinetic energy is lost? A. \(0.03 \%\) B. \(0.3 \%\) C. \(3 \%\) D. \(30 \%\)
-
Louisiana Leisure makes umbrellas, gazebos, and chaise lounges. The company uses a traditional overhead allocation scheme and assigns overhead to products at the rate of $30 per direct labor hour....
-
3) The following divide-and-conquer algorithm performs some mysterious function on an array A[s..t]. The initial call to this recursive function is Find(1, 0, n-1) for an array of n elements. int t)...
-
Consider a uniform mixture of different fluids (all spatial derivatives vanish), with no external forces, such that the equation of motion for the a species becomes (a) Show that the time rate of...
-
Show that the total kinetic energy density of all species in a fluid can be written as the sum of the thermal energy density of the whole fluid plus the kinetic energy of the mass motion, that is...
-
What is the best control procedure to prevent paying the same invoice twice? a. Segregate check-preparation and check-signing functions. b. Prepare checks only for invoices that have been matched to...
-
Go to the Yahoo! Finance website and look up any FTSE 100 company of your choice. Click on the Historical Prices link. Find its closing price yesterday and its closing price exactly one year earlier....
-
In a portfolio of many securities, all having positive correlation with each other, is it possible for the minimum variance portfolio to have zero risk? What happens to the efficient frontier when a...
-
Assume that every asset has the same expected return and variance. Furthermore, all assets have the same covariance with each other. As the number of assets in the portfolio grows, which becomes more...
-
Farrods PLC has a market value of 800 million and 35 million shares outstanding. Redridge department store has a market value of 300 million and 25 million shares outstanding. Farrods is...
-
Classify the following events as mostly systematic or mostly unsystematic: (a) The Bank of Englands base rate increases unexpectedly. (b) A company renegotiates its bank debt after breaching a...
-
The solubility of nitrogen in water is 8.21 10-4 mol/ L at 0oC when the N2 pressure above water is 0.790 atm. Calculate the Henrys law constant for N2 in units of L atm/mol for Henrys law in the...
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
Using any compounds that contain two carbon atoms or fewer, show a way to prepare a racemic mixture of (2R, 3S) - and (2S, 3R) -2, 3-dihydroxypentane.
-
HA has a pK a of 15, while HB has a pK a of 5. Draw the equilibrium that would result upon mixing HB with NaA. Does the equilibrium favor formation of HA or of HB?
-
Explain why each of the following alcohols cannot be prepared via hydroboration-oxidation: a. b. c.
-
1. Title page in APA student format. Refer to Course Resources for APA guides. 2. Article reference (APA format) at the top of the first page 3. Introduction: 1-3 paragraphs orienting the reader to...
-
What forces shaped modern Saudi culture? How similar or different are these forces from those that shaped the culture of Western nations?
-
The Office Costs in the office were getting out of control and the company appointed a new office manager to identify and sort out the problems. Her task was to put together a report for senior...

Study smarter with the SolutionInn App


