A refrigerator using R-410a is powered by a small natural gas-fired heat engine with a thermal efficiency
Question:
A refrigerator using R-410a is powered by a small natural gas-fired heat engine with a thermal efficiency of 25%, as shown in Fig. P9.110. The R-410a condenses at 40◦C and evaporates at −20◦C, and the cycle is standard. Find the two specific heat transfers in the refrigeration cycle. What is the overall COP as QL/Q1?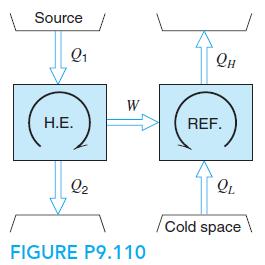
Transcribed Image Text:
Source Q1 Он W Н.Е. REF. Q2 Cold space FIGURE P9.110
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
High temperature 1000K Compressor Work 107 kJkg Turbine Work 162 kJkg The ideal Ericsson cycle consi...View the full answer

Answered By

Firoz K
I have extensive experience in education and tutoring, having worked as a tutor for the past three years in both group and individual settings. During my time as a tutor, I have successfully helped students improve their academic performance in a variety of subjects, including mathematics, science, language arts, and social studies. I have also developed and implemented personalized learning plans and differentiated instruction techniques to accommodate the individual needs of my students. Moreover, I have effectively communicated with parents and teachers to ensure that the students receive the best possible education and guidance. My strong organizational, communication, and problem-solving skills have enabled me to successfully collaborate with students, parents, and teachers in order to provide an effective and enjoyable learning experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A refrigerator using R-22 is powered by a small natural gas fired heat engine with a thermal efficiency of 25%. The R-22 condenses at 40C and it evaporates at-20C and the cycle is...
-
A Carnot heat engine with a thermal efficiency of 60% receives heat from a source at a rate of 3000 kJ/min, and rejects the waste heat to a medium at 300 K. Determine (a) The power that is generated...
-
A heat engine with a thermal efficiency of does 500 J of net work each cycle. How much heat per cycle is lost to the low-temperature reservoir?
-
Olena Mirrors records bad debt using the allowance, income statement method. They recorded $343,160 in accounts receivable for the year and $577,930 in credit sales. The uncollectible percentage is...
-
A unit load AS/RS for work-in-process storage in a factory must be designed to store 2000 pallet loads, with an allowance of no less than 20% additional storage compartments for peak periods and...
-
Write a set of logical predicates that will perform simple automobile diagnostics (e.g., if the engine wont turn over and the lights wont come on, then the battery is bad). Dont try to be too...
-
What role do nitrogen-fixing bacteria and nitrifying bacteria play in the nitrogen cycle?
-
Alamo Foods of San Antonio wants to introduce a new computer system for its perishable products warehouse. The costs and benefits are as follows: a. Given a discount rate of 8 percent (.08), perform...
-
The data structure below is an adjacent matrix that represents a graph. Is this graph cyclic? graph [[ 0, 1, 0, 3], [ 0, 0, 2, 0 ], [ 4, 0, 0, 0], [ 5, 0, 0, 0] ]
-
Duchess Ltd. manufactures and installs kitchen cabinetry. It uses normal job costing with two direct cost categories (direct materials and direct manufacturing labour) and one indirect cost pool for...
-
As explained in the previous problem, the ammonia absorption cycle is very similar to the setup sketched in Problem 9.110. Assume the heat engine has an efficiency of 30% and the COP of the...
-
A split evaporator is used to provide cooling of the refrigerator section and separate cooling of the freezer section, as shown in Fig. P9.109. Assume constant pressure in the two evaporators. How...
-
The accountant for Bannister Co. posted all amounts correctly from the cash receipts journal to the general ledger. However, she failed to post three credits to customer accounts in the accounts...
-
Netflix has been the leader in video-streaming services in the United States since their entry in 2007, outpacing their nearest rival Disney+ and all other competitors. But Netflix has also had a...
-
Question 23 Answer saved Marked out of 1.00 P Flag question Previous page Type here to search A Motor vehicle was purchased on June 1, 2012 for $400 000. It was expected to last for Four years....
-
How do you define deviance , norms , and values ? Give an example of each concept.
-
A collaborative environment can be invigorating and refreshing. This culture also can trickle down to the classroom level where students can benefit too. What are your thoughts?
-
How to test the existence of Heteroskedasticity in the linear regression model? State the Breusch-Pagan test and White test. How to obtain the feasible generalized least square estimator? Explain...
-
Identify some conditions where upper management might allow some divisions to have a lower required rate of return.
-
Name some of the various types of financial intermediaries described in the chapter and indicate the primary reason(s) each was created.
-
Draw the structure of each possible dichloride that can be used to prepare the following alkyne via elimination:
-
Draw the structures of compounds A to D: NaNH, NaNH, 1) Excess NaNH, Br2 Br2 2) H20 (C3H12)
-
Consider the equilibrium 3O 2 (g) 2O 3 (g). a. Using the Data tables, calculate K P at 298 K. b. Assuming that the extent of reaction at equilibrium eq is much less than 1, show that the degree of...
-
2. Two urns contain white and black balls: Urn 1 contains 10 black balls and 5 white balls, Urn 2 contains 8 black balls and 2 white balls. Two balls are drawn randomly from urn 1, and these two...
-
(a) Calculate the inverse Laplace transforms of the following functions. Explain your reasoning. (i) 1-28 482 +36 2s (ii) (iii) (s+3)(s1)2 s+1 82-68+5 (iv) se-48 82-36 [14 marks] (b) Use the Laplace...
-
what ways does the communication of a compelling vision serve to inspire and mobilize individuals and teams towards collective action and goal attainment?

Study smarter with the SolutionInn App


