A steel tank of cross-sectional area 3 m 2 and height 16 m weighs 10 000 kg
Question:
A steel tank of cross-sectional area 3 m2 and height 16 m weighs 10 000 kg and is open at the top, as shown in Fig. P1.59.Wewant to float it in the ocean so that it is positioned 10 m straight down by pouring concrete into its bottom. How much concrete should we use?
In Fig. P1.59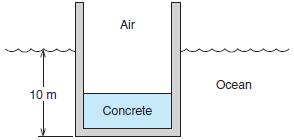
Transcribed Image Text:
Air Ocean 10 m Concrete
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
To determine the amount of concrete needed to float the steel tank in the ocean so tha...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A steel tank of cross sectional area 3 m2 and 16 m tall weighs 10 000 kg and it is open at the top. We want to float it in the ocean so it sticks 10 m straight down by pouring concrete into the...
-
A steel tank of cross sectional area 3 m2 and 16 m tall weighs 10 000 kg and it is open at the top. We want to float it in the ocean so it sticks 10 m straight down by pouring concrete into the...
-
A 5 m long vertical tube of cross sectional area 200 cm2 is placed in a water fountain. It is filled with 15oC water, the bottom closed and the top open to the 100 kPa atmosphere. a) How much water...
-
You are asked to verify Kepler's Laws of Planetary Motion. For these exercises, assume that each planet moves in an orbit given by the vector- valued function r. Let r = ||r||, let G represent the...
-
A lamp costs $32 and is marked up based on cost. If the lamp sold for $72, what was the percent of markup?
-
A fair die is tossed, and the up face is observed. If the face is even, you win $1. Otherwise, you lose $1. What is the probability that you win?
-
The price of a stock share (no dividend, current price is 50) follows a GBM with drift 12% and volatility 35%; the continuously compounded risk free rate is 5%. Consider a (European-style) lookback...
-
You have worked with Zaird & Associates, CPAs, for a little more than a year and are beginning your second audit of Universal Air (UA). This year you even have an assistant reporting to youJane...
-
Moxie Inc., a company that produces typewriter replicas, has fixed costs of $20,000 and variable costs of $18 per unit of output. Their expected unit sales is 10,000 units. What is the unit cost of...
-
At 900oC, Kp = 1.04 for the reaction CaCO3(s) CaO(s) + CO2(g) At a low temperature dry ice (solid CO2), calcium oxide, and calcium carbonate are introduced into a 50.0- L reaction chamber. The...
-
What is the pressure at the bottom of a 5-m-tall column of fluid with atmospheric pressure of 101 kPa on the top surface if the fluid is a. Water at 20C? b. Glycerine at 25C? c. Gasoline at 25C?
-
A piston, mp = 5 kg, is fitted in a cylinder, A = 15 cm 2 , that contains a gas. The setup is in a centrifuge that creates an acceleration of 25 m/s 2 in the direction of piston motion toward the...
-
Panashe Corporation purchased 20 percent of Servco Corporation's stock on each of the following dates: January 4, 2017; April 3, 2017; July 14, 2017; December 15, 2017; and January 5, 2018. Can...
-
What is the name of the gel-like substance, secreted by diverse glands and cells throughout the body, which is prevalent within the respiratory, digestive, and reproductive systems?
-
Financial situation There are 10 branches in your department. 5 of the branches have vote 10 funds totalling $1.1B 2 of the branches have vote 5 funds totalling $500M All of the branches have vote 1...
-
What are the challenges and complexities associated with concurrency in computing systems? Explain
-
Explain market-based solutions to environmental externalities as a public policy decision by drawing a figure that shows imposing tax on pollution and compare the possible effect of this policy to...
-
Q-Sepharose @ pH 3.5, 0-0.5 M Sallt (Lowest pH condition) Q-Sepharose @ pH 11, 0-0.5 M Sallt (Highest pH condition) Q-Sepharose @ pH 7, 0-0.5 M Sallt (Neutral pH condition) Hydrophobic Interaction,...
-
What product(s) would result from nitration of each of the following compounds? a. Propylbenzene b. Bromobenzene c. Benzaldehyde d. Benzenesulfonic acid e. Cyclohexylbenzene f. Benzonitrile
-
A local politician is concerned that a program for the homeless in her city is discriminating against blacks and other minorities. The following data were taken from a random sample of black and...
-
Ammonia in a piston/cylinder arrangement is at 700 kPa, 80C. It is now cooled at constant pressure to saturated vapor (state 2) at which point the piston is locked with a pin. The cooling carries on...
-
A cylinder has a thick piston initially held by a pin as shown in Fig. P3.109. The cylinder contains carbon dioxide at 200 kPa and ambient temperature of 290 K. The metal piston has a density of 8000...
-
Locate the state of ammonia at 200 kPa, -10oC. Indicate in both the P-v and the T-v diagrams the location of the nearest states listed in the printed table B.2
-
Workplace bullying is a phenomenon that has been receiving increasing attention in recent years. Workplace bullying can not only affect the immediate victim but also have ripple effects within the...
-
Ajax Corp's sales last year were $460,000, its operating costs were $362,500, and its interest charges were $12,500. What was the firm's times-interest-earned (TIE) ratio?
-
Nonconstant growth Microtech Corporation is expanding rapidly and currently needs to retain all of its earnings; hence, it does not pay dividends. However, investors expect Microtech to begin paying...

Study smarter with the SolutionInn App


