A400-L tank,A(see Fig. P3.47), contains argon gas at 250 kPa and 30C. Cylinder B, having a frictionless
Question:
A400-L tank,A(see Fig. P3.47), contains argon gas at 250 kPa and 30◦C. Cylinder B, having a frictionless piston of such mass that a pressure of 150 kPa will float it, is initially empty. The valve is opened, and argon flows into B and eventually reaches a uniform state of 150 kPa and 30◦C throughout. What is the work done by the argon?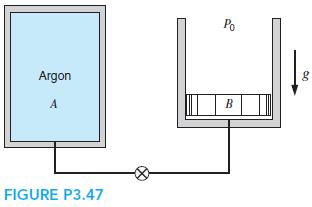
Transcribed Image Text:
Po Argon g A B FIGURE P3.47
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A 3-m3 rigid tank contains hydrogen at 250 kPa and 550 K. The gas is now cooled until its temperature drops to 350 K. Determine (a) The final pressure in the tank and (b) The amount of heat transfer.
-
A piston/cylinder setup contains argon gas at 140 kPa, 10C, and the volume is 100 L. The gas is compressed in a polytropic process to 700 kPa, 280C. Calculate the heat transfer during the process.
-
A 400-L tank, A contains argon gas at 250 kPa, 30C. Cylinder B, having a frictionless piston of such mass that a pressure of 150 kPa will float it, is initially empty. The valve is opened and...
-
Solve the equation (a) Graphically, (b) Numerically, and (c) Symbolically. Then solve the related inequality. |4x7| = 5, |4x - 7| 5
-
Use the test scores of 24 students taking Marketing 235 to complete the frequency distribution and find the grouped mean rounded to the nearest whole number: 57 91 76 89 82 59 72 88 76 84 67 59 77 66...
-
In Problems 9198, use the Venn diagram to indicate which of the eight blood types are included in each set. ANB
-
Find a detailed report on some recent statistical study of interest to you. Write a short report applying each of the eight guidelines given in this section. (Some of the guidelines may not apply to...
-
A trial balance for the Basket Corner, a business owned by Linda Palermo, is shown on page 567. Year-end adjustment information is provided below. (a and b) Merchandise inventory costing $24,000 is...
-
what ways do emerging technologies, such as artificial intelligence and genetic engineering, challenge traditional ethical paradigms, and how should society navigate the ethical dilemmas they present...
-
The data (Prob04-06 in Ch04Data.xlsx on the Student Companion Site) represent the weight of castings (in kilograms) being made in the Harrison Metalwork foundry. Based on this sample of 100 castings,...
-
A 2-kg piston accelerates to 20 m/s from rest. What constant gas pressure is required if the area is 10 cm 2 , the travel is 10 cm, and the outside pressure is 100 kPa?
-
A piston/cylinder assembly contains 2 kg of liquid water at 20C and 300 kPa, as shown in Fig. P3.48. There is a linear spring mounted on the piston such that when the water is heated, the pressure...
-
Describe three ways to conceptualize health policy and law?
-
What are the most effective strategies for fostering self - understanding and personal growth in individuals?
-
Mark Abrahams is a partner at a law firm. He is 40 years old and unmarried. He has incurred a number of expenses during the 2020 year of assessment. He would like to know if they are deductible in...
-
What are the next steps you should follow once you identify the hazards and rate risk levels?
-
Average Stock of Raw Material, WIP, Credit Allowed to debtors, Credit allowed by creditors lag in payment of wages, overheads and Avg finished goods is 1 month 25% of sales are cash sales You may...
-
your 1. (Multiple IRRs) You would like to open an investment company named Rip'em. The cost of building up the company is $750,000. You offer clients the following deal: For five annual deposits of...
-
a. Why is an O--H stretch more intense than an N--H stretch? b. Why is the O--H stretch of a carboxylic acid broader than the O--H stretch of an alcohol?
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
Assume a setup similar to the previous problem but the air flows in the opposite direction of the glass, it comes in where the glass goes out. How much air flow at 17oC is required to cool the glass...
-
Three air flows all at 200 kPa are connected to the same exit duct and mix without external heat transfer. Flow one has 1 kg/s at 400 K, flow two has 3 kg/s at 290 K and flow three has 2 kg/s at 700...
-
Consider the power plant as described in Problem 6.102. a. Determine the temperature of the water leaving the intermediate pressure heater, T13, assuming no heat transfer to the surroundings. b....
-
The COSO framework of internal controls is practiced within companies around the world. The objectives of the COSO framework are closely related to its five components. For this assignment, please...
-
Discuss equipment that is specific to your field of study (Business Administration: Accounting). Use the following guiding questions to stimulate your discussion: What kinds of equipment will you...
-
An individual had the following transactions during 2022 and 2023: item description date proceeds adjusted cost base 1 Sold 1,000 shares of stock A 12 Mar 2022 8,000 6,800 2 Sold 800 shares of stock...

Study smarter with the SolutionInn App


