Assume a Helmholtz equation as where C 0 , C 1 , and C 2 are constants
Question:
Assume a Helmholtz equation as
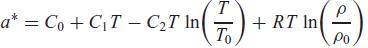
where C0, C1, and C2 are constants and T0 and ρ0 are reference values for temperature and density (see Eqs. 12.92–12.95). Find the properties P, u, and s from this expression. Is anything assumed for this particular form?
Transcribed Image Text:
T + RT In To a* = Co + C| T – C2T In( Po
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The Helmholtz equation represents the thermodynamic potential of a simple compressible substance Giv...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Show that if C1 and C2 are conditions that elements of the n-ary relation R may satisfy, then sC1 (sC2(R)) = sC2 (sC1(R)).
-
Capacitors C 1 and C 2 are in series, with voltage V across the combination. Show that the voltages across the individual capacitors are V 1 = C 2 V/(C 1 + C 2 ) and V2 = C 1 V/(C 1 + C 2 ).
-
Capacitors C1 and C2 are connected in parallel by a resistor and two switches as shown in Figure. Capacitor C1 is initially charged to a voltage V0, and capacitor C2 is uncharged. The switches S are...
-
Let V be the volume of a can of radius r and height h, and let S be its surface area (including the top and bottom). Find r and h that minimize S subject to the constraint V = 54.
-
(b) Lower and upper control limits for the R chart.
-
Which of the following factors are considered when fair use is raised as a defense to a copyright infringement action? a. Nature of the copyrighted work b. Amount and substantiality of the portion...
-
If the frequency is doubled to \(80 \mathrm{kHz}\) and the current is kept the same, what will be the peak out-of-phase voltage? A. \(0.32 \mathrm{~V}\) B. \(0.16 \mathrm{~V}\) C. \(0.080...
-
Leisure Products, Inc., manufactures and sells two products, golf balls and tennis balls. Fixed costs are $100,000, and unit sales are 60,000 sheaths of golf balls and 40,000 cans of tennis balls....
-
Do you feel that the period immediately after the end of the Spanish flu of 1918 provides a good indicator as to how the economy is likely to function in the years of recovery after the COVID-19...
-
Multiple networks connect Quebec and the New-Brunswick and allow transportation to other Canadian provinces and American states. The following graph represent part of that network. We can also see...
-
A new compound is used in an ideal Rankine cycle where saturated vapor at 200C enters the turbine and saturated liquid at 20C exits the condenser. The only properties known for this compound are a...
-
Find an expression for the change in Helmholtz function for a gas with an EOS as P(v b) = R T .
-
Determine the oxidation number of sulfur in each of the following substances: (a) Barium sulfate, BaSO4 (b) Sulfurous acid, H2SO3 (c) Strontium sulfide, SrS (d) Hydrogen sulfide, H2S. (e) Based on...
-
As Paul prepares the budget for his company's sprinkler installation service, he uses DL hours as the allocation base for both variable-MOH and fixed-MOH, since the work is labor-intensive. For a...
-
How can you adjust the following menu items to meet special requests or dietary requirements of customersa) Crumbed chicken schnitzel with salad for a customer with gluten intolerance. b) Creamy rice...
-
Eric is carrying a bucket of golf balls with a total weight of 142.5 ounces. Each ball weighs 1.62 ounces and the bucket alone weighs 21 ounces. Write and solve a linear equation to determine how...
-
A professional concert is played for the benefit of the American Heart Association each year. The admission charge is $32 per ticket, which is also the usual admission price for a professional...
-
Portfolio Planning Problem Following are the investment alternatives available. A: Invest $1 get $1.40 two years later. B: Invest $1 get $1.70 three years later. C: Invest $1 at the beginning of year...
-
a. Give the major product of each of the following reactions: 1. 2. 3. 4. b. What do all the reactions have in common? c. How do all the reactions differ? CH CH Ht CH Ht CH3C-CH2
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
For each pair of compounds below, determine whether they are identical compounds, constitutional isomers, stereoisomers, or different conformations of the same compound: (a) (b) (c) (d) (e) (f) (g)...
-
Predict the major product(s) for each of the following reactions: 1) Hg(OAC)2, -0 2) NABH, ? - O, NaOH, cold Br2 H20 Pt
-
Draw a Lewis structure for each of the following compounds: (a) C 2 H 6 (b) C 2 H 4 (c) C 2 H 2 (d) C 3 H 8 (e) C 3 H 6 (f) CH 3 OH
-
Bridgeport Bus Lines uses the units-of-activity method in depreciating its buses. One bus was purchased on January 1, 2022, at a cost of $221,582. Over its6-year useful life, the bus is expected to...
-
Certified Internal Auditor (CIA) - Certifying body is the Institute of Internal Auditors https://na.theiia.org/certification/CIA-Certification/Pages/CIA-Certification.aspx CIA Certification...
-
Company A has the following data: Prime costs are 180% of direct materials costs. DM costs for year 20xx are $ 100,000. The company had estimated the overhead costs to be $400,000 and the estimated...

Study smarter with the SolutionInn App


