Verify that the ideal gas part of the Helmholtz function substituted in Eq. 12.86 does lead to
Question:
Verify that the ideal gas part of the Helmholtz function substituted in Eq. 12.86 does lead to the ideal gas law, as in the note after Eq. 12.97.
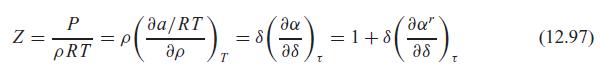
![]()
Transcribed Image Text:
da/RT da" = p pRT ap = 8 as =1+8 a8 (12.97) T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
To verify that the ideal gas part of the Helmholtz function leads to the ideal gas law we can start ...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Assuming that the ideal gas law holds, find the amount of nitrogen gas in a container if P = 0.836 atm 0.003 atm, V = 0.01985 m 3 0.00008 m 3 , T = 298.3 K 0.2 K. Find the expected error in the...
-
The ideal gas law relates four variables. An empirical gas law relates two variables, assuming the other two are constant. How many empirical gas laws can be obtained? Give statements of each.
-
The gas law for an ideal gas at absolute temperature T (in kelvins), pressure P (in atmospheres), and volume V (in liters) is PV = nRT, where is the number of moles of the gas and R = 0.0821 is the...
-
A machine fills containers with a particular product. The standard deviation of filling weights computed from past data is 0.6 ounces. If only 2% of the containers hold less than 18 ounces, what is...
-
Suppose 1 = $0.0077 in London, $1 = SF2.00 in New York, and SF1= 65 in Paris. a. If you begin by holding 10,000 yen, how could you make a profit from these exchange rates? b. Find the arbitrage...
-
Water forms beads on the surface of a newly painted car. After the car is exposed to the weather for a long time, water spreads out into a thin film on its surface. What has happened to the paint to...
-
How do bacteria reproduce? Do bacteria ever exchange genetic material?
-
Consider the budgeted income statement for Carlson Company for June 20X4 in Exhibit 7-13. The cash balance, May 31, 20X4, is $15,000. Sales proceeds are collected as follows: 80% the month of sale,...
-
Consider the sensitivity report below for the problems which follow. variable cells cell name final value reduced cost objective coefficient allowable increase allowable decrease $b2 units produced...
-
X Ltd. has 10 lakhs equity shares outstanding at the beginning of the accounting year 2016. The appropriate P/E ratio for the industry in which D Ltd. is 8.35. The earnings per share is Rs. 15 in the...
-
R-410a is a 1:1 mass ratio mixture of R-32 and R-125. Find the specific volume at 20C, 1200 kPa, using Kays rule and the generalized charts, and compare it to the solution using Table B.4.
-
Gases like argon and neon have constant specific heats. Develop an expression for the ideal gas contribution to the Helmholtz function in Eq. 12.92 for these cases. a* = h* RT Ts* (12.92)
-
Describe how the BCG matrix and diversification are used to establish corporate-level strategy.
-
What is organizational agility and why is it important? (elaborate) Literature: Agility is a complex construct that can take multiple forms. It captures an organization's ability to develop and...
-
What is the prevailing stance among contemporary sociologists regarding the nature versus nurture debate?
-
Consider total cost function: C(q) = 48 + 3q2 + 2q, derive the average cost function, the marginal cost function, and the minimum efficient scale, and carefully graph the average cost and marginal...
-
PROBLEMS: ACCOUNTS RECEIVABLES 7. Effective with the year ended December 31, 2023, Vega Company adopted the aging of accounts receivable method instead of the old percentage of sales method. Vega has...
-
A 350-kg piano slides 3.6 m down a 22 incline and is kept from accelerating by a man who is pushing back on it parallel to the incline.(Figure 1) Determine the force exerted by the man. Express your...
-
What are the three lowest angular speeds for which the wheel in Fizeau's apparatus (see Fig. 22.13) allows the reflected light to pass through to the observer? Assume the distance between the notched...
-
Should we separate the debt and equity features of convertible debt? Team 1: Pro separation: Present arguments in favor of separating the debt and equity features of convertible debt. Team 2: Against...
-
Rank the following compounds in terms of increasing basicity: N. Br z-
-
When (E)-4-amino-3-buten-2-one is treated with molecular hydrogen in the presence of platinum, the resulting amine is more basic than the reactant. Draw the reactant and the product, and explain why...
-
For each of the following compounds, draw the form that predominates at physiological pH: (a) (b) (c) CI CH CI Sertraline (Zoloft) An antidepressant NH2 Amantadine Used in the treatment of...
-
Katie has decided to move forward with attaining the property from the Week 1 Case Analysis. However, she is not certain how to go about financing the property, either through a short sale offer or...
-
Bonds are a vital source of financing for corporations and governments. Bonds are rated based on their default risk. Some bonds, such as Treasury bonds, have no risk of default, whereas so-called...
-
List customer ID, customer name, order ID, order date, product ID and the quantity of each product ordered, for all customers who have placed orders. Hint: You need to include 3 tables in your FROM...

Study smarter with the SolutionInn App


