A 2.5-cm-diameter horizontal water jet with a speed of V j = 40 m/s relative to the
Question:
A 2.5-cm-diameter horizontal water jet with a speed of Vj= 40 m/s relative to the ground is deflected by a 60? stationary cone whose base diameter is 25 cm. Water velocity along the cone varies linearly from zero at the cone surface to the incoming jet speed of 40 m/s at the free surface. Disregarding the effect of gravity and the shear forces, determine the horizontal force F needed to hold the cone stationary.
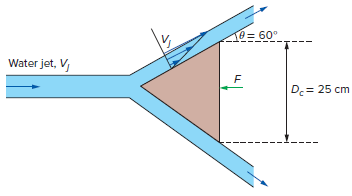
T0= 60° Water jet, V D.= 25 cm
Step by Step Answer:
A horizontal water jet is deflected by a stationary cone The ho...View the full answer



Fundamentals of Thermal-Fluid Sciences
ISBN: 978-0078027680
5th edition
Authors: Yunus A. Cengel, Robert H. Turner, John M. Cimbala
Related Video
In Physics, a force is an influence that can change the motion of an object. A force can cause an object with mass to change its velocity, i.e., to accelerate. Force can also be described intuitively as a push or a pull. A force has both magnitude and direction, making it a vector quantity. In this video, the concept of force and torque is demonstrated with the practical application of changing the flat tire of the car.
Students also viewed these Physics questions
-
A 12-cm diameter horizontal water jet with a speed of V j = 25 m/s relative to the ground is deflected by a 40° cone moving to the left at V c = 10 m/s. Determine the external force, F, needed to...
-
A 3-in-diameter horizontal water jet having a velocity of 90 ft/s strikes a curved plate, which deflects the water 180? at the same speed. Ignoring the frictional effects, determine the force...
-
A jet plane is flying at 130 m/s relative to the ground. There is no wind. The engines take in 81 kg of air per second. Hot gas (burned fuel and air) is expelled from the engines at high speed. The...
-
Consider a stylized two-period model with banking. The aggregate abatement cost function in period t is given by C(E)= (a t be) 2 /2b with a 1 < a 2 is D(E)=dE 2 /2. (a) Determine the optimal...
-
When the bulb of a constant-volume gas thermometer is placed in a beaker of boiling water at 100 °C, the pressure of the gas is 227 mmHg. When the bulb is moved to an ice-salt mixture, the...
-
Limonene is a compound found in orange oil and lemon oil. When limonene is treated with excess hydrogen and a platinum catalyst, the product of the reaction is 1-isopropyl-4-methylcyclohexane. When...
-
Use stepwise regression to build a model for the NBA Combine data in Table B.23. Perform a residual analysis on the final model. Compare this model to the all possible regressions model from Problem...
-
Kleen Company acquired patent rights on January 10 of Year 1 for $2,800,000. The patent has a useful life equal to its legal life of eight years. On January 7 of Year 4, Kleen successfully defended...
-
Osprey Co. prepared a master budget for 2021 reflecting indirect material costs of $163,842 based on annual production of 127,179 widgets (the annual production is spread out equally over the twelve...
-
Are the following functions analytic? Use (1) or (7). f(z) = Arg 2z
-
A horizontal water jet of constant velocity V impinges normally on a vertical flat plate and splashes off the sides in the vertical plane. The plate is moving toward the oncoming water jet with...
-
Water enters a 10-cm-diameter pipe steadily with a uniform velocity of 3 m/s and exits with the turbulent flow velocity distribution given by u = u max (1 - r/R)1 /7 . If the pressure drop along the...
-
Marie Corporation manufactures a fiber optic connector. The variable cost per unit is $16. The fixed cost per unit is $9. The companys desired ROI per unit is $3. Compute the markup percentage using...
-
Enter your answer in the provided box. Hydrogen sulfide decomposes according to the following reaction, for which K = 9.30 108 at 700C: 2 HS(g) 2 H(g) + S(g) If 0.49 mol of HS is placed in a 3.0-L...
-
How many molecules of N2O4 are in 76.8 g of N2O4? O 4.99 X 1024 molecules of N2O4 O 1.38 X 1024 molecules of N2O4 O 4.59 X 1025 molecules of N2O4 O5.03 X 1023 molecules of N2O4
-
What are the angles a and b in the actual molecule of which this is a Lewis structure? H a C H H H a Note for advanced students: give the ideal angles, and don't worry about small differences from...
-
8. What is the [H0] in a solution of 0.0005 M NaOH? 9. What is (OH] in a 0.00240 M solution of the strong acid HBr? 10. A commercial window-cleaning liquid has a pH of 11.7. What is the hydroxide ion...
-
Monochlorination of pentane, C5H12, leads to formation of how many different products? 2 O1 05
-
Four projects are available for investment. The projects require the cash flows and yield the net present value s (in millions) shown in the file P06_39.xlsx. If $6 million is available now for...
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
Three identical 6.40-kg masses are hung by three identical springs, as shown in Fig. E6.33. Each spring has a force constant of and was 12.0 cm long before any masses were attached to it. (a) Draw a...
-
A 2.0-kg box and a 3.0-kg box on a perfectly smooth horizontal floor have a spring of force constant 250 N/m compressed between them. If the initial compression of the spring is 6.0 cm, find the...
-
A 6.0-kg box moving 3.0 m/s at on a horizontal, frictionless surface runs into a light spring of force constant 75 N/cm. Use the workenergy theorem to find the maximum compression of the spring?
-
For each ordered pair, determine -6x+7y=2 7x-4y=6 a solution to the system of equations.
-
Review the meaning of the concepts or terms given in Key Terms and Concepts. Distinguish between a production department and a service department. Distinguish between a direct cost and an indirect...
-
What would you like the members of your learning community to know about your global health interests, perspectives, and life experiences? How will you prepare to listen and learn from others? Why...

Study smarter with the SolutionInn App


