The following 2.5 m 8.1 m 6 m tank shown is filled by oil of
Question:
The following 2.5 m × 8.1 m × 6 m tank shown is filled by oil of SG = 0.88. Determine
(a) The magnitude and the location of the line of action of the resultant force acting on surface AB
(b) The pressure force acting on surface BD. Will the force acting on surface BD equal the weight of the oil in the tank? Explain.
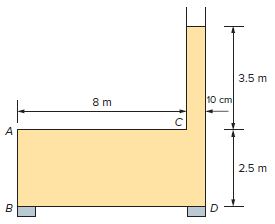
3.5 m 8 m 10 cm A 2.5 m B D
Step by Step Answer:
A tank is filled by oil The magnitude and the location of the line of action of th...View the full answer



Fundamentals of Thermal-Fluid Sciences
ISBN: 978-0078027680
5th edition
Authors: Yunus A. Cengel, Robert H. Turner, John M. Cimbala
Related Video
As we know, to break a coconut we have to apply 400 pounds of pressure to it. In pounds = 400psi In newton = 1779.29N Starting from the first person he will try to break the coconut with his hands. As we know humans can exert a force 292 newtons by their hands. So force by hand is approximately = 300 N Then the second and third one also try to break it by hand but they fail too.
Students also viewed these Physics questions
-
Wastewater containing volatile species A dissolved in the water at a dilute inlet concentration of 0.50 gmole A/m 3 (c AL,0 ) is pumped into a well-mixed open holding pond at a volumetric flow rate...
-
(a) [COCI2] = 2.00 Ã 10-3 M, [CO] = 3.3 Ã 10-6 M, [CI2] = 6.62 Ã 10-6 M; [CI2] = 6.62 Ã 10-6 M; (b) [COCI2] = 4.50 Ã 10-2 M, [CO] = 1.1 Ã 10-7 M, [CI2] =...
-
The absorption cross section on the ordinate of the ozone absorption spectrum at the beginning of this chapter is defined by the relation where n is the number of absorbing molecules per cubic...
-
Suppose there are two identical forest plots except that one will be harvested and left as is while the second will be cleared after the harvest and turned into a housing development. In terms of...
-
Water flows through a horizontal tube of diameter 2.5 cm that is joined to a second horizontal tube of diameter 1.2 cm. The pressure difference between the tubes is 7.3 kPa. (a) Which tube has the...
-
(a) Outline a Kiliani-Fischer synthesis of epimeric aldopentoses starting with D-(-)- erythrose (use Fischer projections). (b) The two epimeric aldopentoses that one obtains are D-(-)-arabinose and...
-
Consider the model \[ y=\theta_{1}-\theta_{2} e^{-\theta_{3} x}+\varepsilon \] This is called the Mitcherlich equation, and it is often used in chemical engineering. For example, \(y\) may be yield...
-
Create a context-level diagram for billing in a dental office. External entities include the patients and insurance companies.
-
Print Company acquired 80% of the stock of Sand Corp. on January 1, 2023. The stockholder's equity section of Sand's balance sheet at that date is as follows: Stockholder's Equity Section Common...
-
A database has four transactions. Let min sup 60 % and min conf 80 % cust ID TID items.bought (in the form of brand-item.category) T100 King's-Crab, Sunset-Milk, Dairyland-Cheese, Best-Bread T200...
-
From Prob. 11-22, knowing that the density of the suspension depends on liquid depth and changes linearly from 800 kg/m3 to 900 kg/m3 in the vertical direction, determine the resultant force acting...
-
The two sides of a V-shaped water trough are hinged to each other at the bottom where they meet, as shown in Fig. P11??25, making an angle of 45? with the ground from both sides. Each side is 0.75 m...
-
Prove that the tangent of the angle (0 /2) between the radial line and the tangent line at the point (r, ) on the graph of r = () (see figure) is given by tan = dr/de R|N 2 Polar curve: r = f(0)...
-
Would a polar protic solvent like ethanol favor the solvolysis of 2-chloro-2-methylpropane? Why or why not? View Available Hint(s) O Ethanol would favor the solvolysis; the mechanism is E1. O Ethanol...
-
Prepare a 0.15% (w/v) solution of haemoglobin in 10 mM HCl pH2. Dilute to 0.03, 0.05, 0.07% and 0.09% in 10 mM HCl pH 2. 2. Prepare a calibration curve of haemoglobin in 10mM HCl pH 2 at 372 nm...
-
What happens to the pressure of a sample of gas if it is allowed to exp three times its original volume? The temperature remains constant. Pressure is reduced by a third Pressure is halved Pressure...
-
3. Benzoic acid + methanol III. Write the equation for the acid hydrolysis of the following esters: 0 11 1. H -C-0-CHz - CH3 2. Ethyl butanoate heat 3. Methyl propanoate Page
-
An experimental procedure for standardization of the titrant states the following: Weigh accurately, about 0.26-0.27 g of the primary-standard. Record the mass to the nearest 0.0001 g. Which mass...
-
Based on Angel et al. (2003). A fertilizer company is trying to determine the cheapest fertilizer mix that provides desired amounts of nutrients. The mix is made by combining the following...
-
Which of the followingcarbocations is the least stable? CH3CH2 . CH3CHCH3 CH3 I . CH3C0 T CH3 IV. V. CH3 CH3CCH2 CH3
-
A particle moving along the x-axis has its velocity described by the function v x = 2t 2 m/s, where t is in s. Its initial position is x 0 = 1 m at t 0 = 0 s. At t = 1 s what are the particles (a)...
-
The position of a particle is given by the function x = (2t 3 - 9t 2 + 12) m, where t is in s. a. At what time or times is v x = 0 m/s? b. What are the particles position and its acceleration at this...
-
The position of a particle is given by the function x = (2t 3 6t 2 + 12) m, where t is in s. a. At what time does the particle reach its minimum velocity? What is (v x ) min ? b. At what time is the...
-
Siran plans to contribute $850 at the start of every half year to an investment that ears 7% compounded monthly. If Siran starts contributing on his 37th birthday, how much will he accumulate by his...
-
Milwaukee Insurance Company (MIC) has entered into a four-year plain vanilla swap with a counterparty. The notional principal is $80 million. MIC will pay payments based on a floating rate of the...
-
Newton Bay is a sparsely populated area. The government plans to build an international airport there and announces this plan publicly. When hotel chains learn about the airport, they become...

Study smarter with the SolutionInn App


