According to the Lewis theory, each of the following is an acidbase reaction. Which species is the
Question:
According to the Lewis theory, each of the following is an acid–base reaction. Which species is the acid and which is the base?
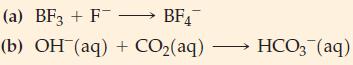
Transcribed Image Text:
BF4 (a) BF3 + F- (b) OH(aq) + CO₂(aq) HCO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Analyze Recall that in Lewis theory an acidbase reaction involves the movement of electrons The Lewi...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
The three following reactions are acidbase reactions according to the Lewis theory. Draw Lewis structures, and identify the Lewis acid and Lewis base in each reaction. (a) B(OH)3 + OH (b) NH4 + H3O+...
-
Define an acid and a base according to the Lewis concept. Give a chemical equation to illustrate.
-
DBU Systems manufactures testing equipment for the communications industry. In developing a new device for maritime communication, the design group has estimated the following unit costs. Metal...
-
Refer to Exhibit 16-2, which contains the PepsiCo Inc. listing from Mergents Handbook of Dividend Achievers. Assume that an investor has asked you to assess PepsiCos recent history and prospects....
-
A $1,000 bond has a coupon rate of 10 percent and matures after eight years. Interest rates are currently 7 percent. a) What will the price of this bond be if the interest is paid annually? b) What...
-
For approximately 20 months, Robert E. McDonald perpetrated a scheme to solicit millions of dollars purportedly for a \($100\) million purchase by the RAI Entities and certain other related corporate...
-
Picard Landscaping plants grass seed as the basic landscaping for business campuses. During a recent month, the company worked on three projects (Remington, Chang, and Wyco). The company is...
-
As part of a survey, 15 adults were asked, "How many hours did you spend at your job last week?" The results are shown in the stem-and-leaf display below. Use the display to answer the questions that...
-
(A) The solubility of CO 2 (g) in H 2 O at 25 C and under a CO 2 (g) pressure of 1 atm is 1.45 g CO 2 /L. Air contains 0.037% CO 2 by volume. Use this information, together with data from Table 16.5,...
-
(A) Sodium fluoride, NaF, is found in some toothpaste formulations as an anticavity agent. What is the pH of 0.10 M NaF(aq)? (B) The pH of an aqueous solution of NaCN is 10.38. What is [CN - ] in...
-
In what funds might property and other nonfinancial resources be recorded?
-
Inez Company has the following target capital structure: 35% debt, 55% common stock, and 10% preferred stock. The company's before-tax cost of debt is 6%, its cost of common stock is 10%, and its...
-
AmBev is a large Brazilian beverage company. You estimate the next year's FFCF at $10.5 billion. You also estimate the perpetual growth at 3.4%. It is a 100% equity funded company with a market...
-
An investor has reviewed a business plan and the company's track record since its inception six months ago. The investor is ready to provide capital for the company to grow. Why is the investor...
-
Bro Partners is a general partnership formed in 2018. The business provides IT services to customers. In 2020 the business made a profit for the first time. When tax time comes around the partnership...
-
A technician from India continues to receive their Mumbai base salary and is kept on the operation's balance sheet. The company then adds various allowances for cost of living, housing, education,...
-
The warrants of Integra Life Sciences allow the holder to buy a share of stock at $11.75 and are selling for $2.85. The stock price is currently $8.50. To what price must the stock go for the warrant...
-
Is it ethical to provide safety training in English to immigrant workers who speak little English, in order to reduce costs?
-
On January 1 Kwun Corporation purchased a 25% equity in Connors Corporation for $180,000. At December 31 Connors declared and paid a $60,000 cash dividend and reported net income of $200,000....
-
Presented below are two independent situations. 1. Heath Cosmetics acquired 15% of the 200,000 shares of common stock of Van Fashion at a total cost of $13 per share on March 18, 2010. On June 30,Van...
-
Ryan Company purchased 70% of the outstanding common stock of Wayne Corporation. Instructions (a) Explain the relationship between Ryan Company and Wayne Corporation. (b) How should Ryan account for...
-
what are 40 companies that may need financial analyst positions?
-
Your supervisor sees your potential as a future analyst for a large brokerage firm. (You haven't told her you want to work for her firm after graduating. But that's okay; everything will be in due...
-
Is the correct answe "sharpe uses standard deviation for the denominator and sortino uses the downside deviation for the denominator?

Study smarter with the SolutionInn App


