Determine K c for the reaction 2 N2(g) + O2(g) + Br2(g) NOBr(g) from the
Question:
Determine Kc for the reaction
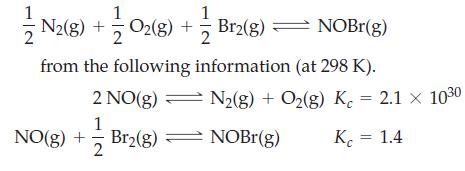
Transcribed Image Text:
2 N2(g) + O2(g) + — Br2(g) — NOBr(g) from the following information (at 298 K). 2 NO(g) 1 Br₂(g) NO(g) + 2 N2(g) + O₂(g) K = 2.1 × 10³⁰ NOBr(g) K = 1.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine Kc for the reaction we can use the following equation Kc productsn reactantsm where n a...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
How is HRM technology being used to "strategically" recruit and staff? Share an example. How does screening software present challenges for recruitment and staffing? How does staffing HRM technology...
-
The equilibrium constant Kc for the reaction is 3.8 Ã 10-5 at 727°C. Calculate Kc and KP for the equilibrium at the same temperature. 12(g)--21(g) 21(g)- 2(g)
-
The equilibrium constant Kc for the reaction is 2.18 Ã 106 at 730°C. Starting with 3.20 moles of HBr in a 12.0-L reaction vessel, calculate the concentrations of H2, Br2, and HBr at...
-
Joe rents his condo for $1,500 per month. Total rental and personal use days for the current year was 210 days and 20 days, respectively. What are the tax consequences for Joe?
-
Jenny Rene, the CFO of Asor Products, Inc., has just completed an evaluation of a proposed capital expenditure for equipment that would expand the firms manufacturing capacity. Using the traditional...
-
Anthony and Karen were partners doing business as the Petite Garment Company. Leroy owned a dye plant that did much of the processing for the company. Anthony and Karen decided to offer Leroy an...
-
Africa offers a fast growing and exciting mobile phone market, with more than 50% growth per year since 2002. More than 28% of African consumers own mobile phones; a larger market than North America....
-
A committee in charge of promoting a Ladies Professional Golf Association tournament is trying to determine how best to advertise the event during the two weeks prior to the tournament. The committee...
-
(20%) The output of an argon ion laser can consist of a number of modes of frequency that match the cavity resonance condition and are within the gain bandwidth of the lasing transition. Assume the...
-
A 0.0240 mol sample of N 2 O 4 (g) is allowed to come to equilibrium with NO 2 (g) in a 0.372 L flask at 25 C. Calculate the amount of N 2 O 4 present at equilibrium (Fig. 15-9). Figure 15-9 NO4(g) 2...
-
Ammonium hydrogen sulfide, NH 4 HS(s), used as a photographic developer, is unstable and dissociates at room temperature. A sample of NH 4 HS(s) is introduced into an evacuated flask at 25 C. What is...
-
Explain three factors that will influence the balance of online and offline web site promotion for an organization.
-
Do you have experience using an HRIS System (Peoplesoft, Workday, Banner, UKG, etc.) to input, update, review and analyze employee and organizational data. Be sure to identify the job(s) in your...
-
The Field, Brown & Snow are partners and share income and losses equality. The partner decide to liquidate the partnership when their capital balances are as follows: Field, $131,250; Brown,...
-
Write a program that reads two integers from the user then dis- plays their sum, product, difference, quotient and remainder.
-
Given the demand curve, O = 100 - 2P. Find the elasticity of demand if the price increase from $20 to $25 using the arch or midpoint elasticity formula. Between the price change from $20 to $25, will...
-
Norman Company has an opportunity to produce and sell a revolutionary new smoke detector for homes, and the project would expect to last for 6 years. To determine whether this would be a profitable...
-
Meera Corporation's comparative balance sheets are presented below. Additional information: 1. Net income was $22,766. Dividends declared and paid were $17,716. 2. All other changes in noncurrent...
-
Do public and private companies follow the same set of accounting rules? Explain.
-
Staircase Equipment Company uses a job order cost system. The following data summarize the operations related to production for April 2010, the first month of operations: a. Materials purchased on...
-
Lynch Furniture Company refinishes and reupholsters furniture. Lynch uses a job order cost system. When a prospective customer asks for a price quote on a job, the estimated cost data are inserted on...
-
Dacher Company uses a job order cost system. The following data summarize the operations related to production for October: a. Materials purchased on account, $450,000. b. Materials requisitioned,...
-
= 4. An alpha particle, (Q = +2e) with kinetic energy Ki 5.30 MeV happens to be headed directly toward the nucleus of a neutral gold atom, (QAu = +79e). What is its distance of closest approach d...
-
Using the Quick Fix tool, create WolfScheduler's public methods from the compiler errors in WolfSchedulerTest. You can adjust the parameter names as you work on implementing each method in later...
-
1. What is the difference between a conventional loan and an insured mortgage loan? 2. What is the loan-to-value ratio? 3. How does a FHA loan works, including how much down payment the borrower must...

Study smarter with the SolutionInn App


