Determine values of K c from the K p values given. (a) SOCl(g) SO(g) + Cl(g) (b)
Question:
Determine values of Kc from the Kp values given.
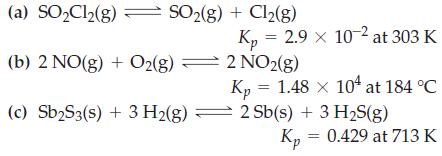
Transcribed Image Text:
(a) SO₂Cl₂(g) SO₂(g) + Cl₂(g) (b) 2 NO(g) + O2(g) (c) Sb₂S3(s) + 3 H₂(g) Kp = 2.9 x 10-2 at 303 K 2 NO2(g) Kp = 1.48 × 104 at 184 °C 2 Sb(s) + 3 H₂S(g) Kp = 0.429 at 713 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
the values of Kc from the Kp values given a SO2Cl2gSO2g C...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
What is a bureaucrat? Do they operate within a framework that is different from the private sector? If so, explain the differences. What is the effect of the bureaucrat and/or the bureaucracy on the...
-
For the reaction show that Kc = Kp /(RT) Do not use the formula Kp = Kc(RT)n given in the text. See Problem 14.99. COCI2(g) CO(g) + Cl2(g)
-
The equilibrium constant Kc for the reaction is 3.8 Ã 10-5 at 727°C. Calculate Kc and KP for the equilibrium at the same temperature. 12(g)--21(g) 21(g)- 2(g)
-
Who was the petitioner? Who was the respondent? b. In what year was the case heard? c. What tax years did the case address? d. Who was the judge in the case? e. What was the basic issue in the case?...
-
Etsitty Arts, Inc., a leading producer of fine cast silver jewelry, is considering the purchase of new casting equipment that will allow it to expand the product line into award plaques. The proposed...
-
Robert applies to Northern National Bank for a loan. Prior togranting the loan, Northern requests that Callis Credit Agency provide it with a credit report on Robert. Callis reports that three years...
-
In question 1, identify the marketing as opposed to the business strategy. Data From Question 1: What is a business strategy? Do you agree with the definition proposed? Illustrate your answer with...
-
Jannero Pargo opened Pargos Cleaning Service on July 1, 2012. During July the following transactions were completed. July 1 Pargo invested $20,000 cash in the business. 1 Purchased used truck for...
-
3. A 200 kg roller coaster starts from rest at the top of the first hill at a height of 20 m above the ground. The second hill is 15 m above the ground. a. A physics student in line for the ride...
-
After the hypothetical reaction A(g) + B(g) C(g) reaches equilibrium in a closed container, 0.100 mol of the inert gas argon is added. In addition, the volume of the container is decreased....
-
(A) The reaction N 2 O 4 (g) 2 NO 2 (g) is at equilibrium in a 3.00 L cylinder. What would be the effect on the concentrations of N 2 O 4 (g) and NO 2 (g) if the pressure were doubled (that is,...
-
How is a cash budget different from a set of pro forma financial statements? Why do you think that firms typical create cash budgets at higher frequencies than they create pro forma statements?
-
-1 Reverse osmosis membrane filtration is to be used to treat brackish (saline) groundwater to supply drinking water for a NSW regional city. Feed water will be provided at a flow rate of 25,000...
-
The following data applies to GoodCorp. (in millions of US $): Cash & marketable securities $315 Fixed assets $546 Net sales $2520 Earnings Before Interests and Taxes (EBIT) $273 Net Earnings After...
-
Lee Inc. is an American firm that will need to make a payment of 889,000 Australian dollars in 90 days. Currently, the spot exchange rate is USD 0.9377/AUD. The company wants to hedge the transaction...
-
A comet collided with Planet X. Assume that these fragments came from an infinite distance away with zero initial speed and fell into planet x. What was there speed when they arrived at planet x's...
-
What should you do after finishing work with a reagent bottle? Select one or more: Return the bottle to its proper storage location. Close the bottle. Place the bottle in the Solid Waste container....
-
What are some of the challenges to developing a system of communications based on wireless media?
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Prepare a cost of production report for the Cutting Department of Perma-Wear Carpet Company for October 2010, using the following data and assuming that all materials are added at the beginning of...
-
Performance Castings Inc. casts blades for turbine engines. Within the Casting Department, alloy is first melted in a crucible, then poured into molds to produce the castings. On December 1, there...
-
Franklin Paper Company manufactures newsprint. The product is manufactured in two departments, Papermaking and Converting. Pulp is first placed into a vessel at the beginning of papermaking...
-
3) Find the moment of inertia of a hollow cylinder with mass m, length L. The object is rotating around its axis of symmetry. The distance from the axis to the inner edge of the shell is a, and the...
-
2] Consider the following system of spring-mass subject to forced vibration: k m C the Governing differential this system THI & my + cj + by = fit) governing differential solve the f(+) = { ++ 1/2 - n
-
Two masses m = 22 kg and mB = 4.0 kg are on inclines and are connected together by a string as shown in (Figure 1). The coefficient of kinetic friction between each mass and its incline is = 0.30....

Study smarter with the SolutionInn App


