For each of the following reactions, predict the products and the mechanisms by which the products are
Question:
For each of the following reactions, predict the products and the mechanisms by which the products are formed.
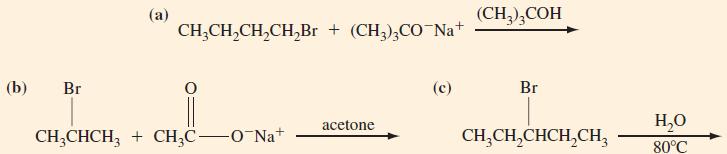
Transcribed Image Text:
(b) Br (a) CH₂CH₂CH₂CH₂Br + (CH3)3CO-Na+ CH3CHCH3 + CH₂C-O-Na+ acetone (c) (CH3)3COH Br H₂O CH,CH,CHCH,CH, 80°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Analyze We follow the sixstep approach outlined above and use the decision tree in Figure 2713 to guide our thinking Figure 2713 Solve a We identify t...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Each of the following reactions has been reported in the chemical literature and proceeds in good yield. What are the principal organic products of each reaction? In some of the exercises more than...
-
how many mono bromination products would be out what product (s) would you expect from each of the following reactions below in each part, give the mechanisms (sn1, sn2, e1, or e2) through which each...
-
Each of the following reactions has been reported in the chemical literature and proceeds in good yield. What are the principal organic products of each reaction? In some of the exercises more than...
-
Find the volumes of the solids generated by revolving the regions bounded by the lines and curves about the x-axis. The region between the curve and the x-axis from x = 1/4 to x = 4 y = 1/(2x)
-
Heink Company incurred $32,056 in manufacturing overhead and applied $32,537. The company uses the most common and simple method of handling differences between actual and applied overhead. Make the...
-
There are corporate goals other than to improve profit. Name three.
-
Why is it advisable to obtain a written confession from the subject of an investigation?
-
Southwestern University (SWU), located in the small town of Stephenville, Texas, is experiencing increased interest in its football program now that a big-name coach has been hired. The increase in...
-
By how much does the fluid level rise in the side of the manometer that is open to the atmosphere? By how much does the fluid level rise in the side of the manometer that is open to the atmosphere if...
-
Write a balanced chemical equation for the reaction that is described and then classify the reaction as a substitution, an elimination, an addition, or a rearrangement reaction: (a) Ethene and Br 2...
-
Would hydration of (E)-but-2-ene yield exclusively (R)-butan-2-ol, (S)-butan-2-ol, or a mixture of the (R) and (S) enantiomers? Explain.
-
In the following reactions indicate which is the Lewis acid and which is the Lewis base: (a) SOI2 + BaSO3 (b) HgCl3 + CI - 2+ Ba+ + 21 +2SO HgC14-
-
What are the units of k in the following rate law? Rate = k [ x ] 2 [ Y ] A ) 1 M 2 s 2 B ) 1 M 2 s C ) M 2 s D ) M s E ) 1 M 3 s
-
You used your 10mL graduated cylinder to measure out acetic anhydride. Now you need to obtain 10mL of methanol. You wash the graduated cylinder and place it in the drying oven for a few minutes to...
-
A 840.3 ng sample of an unknown radioactive substance was placed in storage and its mass measured periodically. After 47 days the amount of radioactive substance had decreased to 105.0 ng. How many...
-
When measuring volume from a 5mL graduated pipette, the volume should be reported in significant figures. 0 1 2 O 3 10 pts 4
-
Particle accelerators can impart very high energies to charged particles like electrons. What is the wavelength ( in nm ) of an electron ( mass = 9 . 1 0 9 4 x 1 0 ^ - 3 1 kg ) that has been...
-
What are the two major functions of administrative agencies?
-
1. Firms may hold financial assets to earn returns. How the firm would classify financial assets? What treatment will such financial assets get in the financial statements in accordance with US GAAP...
-
Barnes cash budget for the entire year, although not given here, is based heavily on his forecast for monthly sales. Sales are expected to be extremely low between May and September but then increase...
-
What reasons might SKI have for maintaining a relatively high amount of cash?
-
Is there any reason to think that SKI may be holding too much inventory? If so, how would that affect EVA and ROE?
-
Required Each of the following independent events requires a year-end adjusting entry. Show how each event and its related adjusting entry affect the accounting equation. Assume a December 31 closing...
-
Moody Corporation uses a job-order costing system with a plantwide predetermined overhead rate based on machine-hours. At the beginning of the year, the company made the following estimates:...
-
Swifty Corporation had the following assets and liabilities on the dates indicated. December 31 Total Assets Total Liabilities 2021 $469,000 $216,000 2022 $549,000 $266,000 2023 $679,000 $366,000...

Study smarter with the SolutionInn App


