For the reaction answer the following questions: (a) Can a solution be prepared at 298 K that
Question:
For the reaction
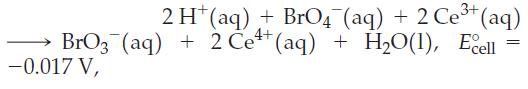
answer the following questions:
(a) Can a solution be prepared at 298 K that has
![= [BrO4] = [Ce4+] = 0.675 M, [BrO3] = [Ce+] = 0.600 M and pH = 1?](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/8/1/1/8856560546daabf92.png)
(b) If not, in which direction will a reaction occur?
Transcribed Image Text:
2 H+ (aq) + BrO4 (aq) + 2 Ce³+ (aq) BrO3(aq) + 2 Ce+ (aq) + H₂O(1), Ecell -0.017 V, =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Given the cell reaction 2 Haq BrO4aq 2 Ce aq 3 Brosaq 2 Ceaq H2Ol The standard cell potential Ecell ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
For the reaction 2 Cu + (aq) + Sn 4+ (aq) 2 Cu 2+ (aq) + Sn 2+ (aq), E cell = -0.0050 V, (a) Can a solution be prepared at 298 K that is 0.500 M in each of the four ions? (b) If not, in which...
-
In which direction will the position of the equilibrium 2HI(g) H2(g) + I2(g) be shifted for each of the following changes? a. H2(g) is added. b. I2(g) is removed. c. HI(g) is removed. d. In a rigid...
-
In the text the equation G = Go + RT ln(Q) was derived for gaseous reactions where the quantities in Q were expressed in units of pressure. We also can use units of mol/ L for the quantities in Q...
-
A company with a seasonal demand for its products may have an opportunity to obtain an off-season order. The relevant factors for such a short-run decision include the a. additional revenues and...
-
Review the basic areas of advertising regulation. Are such regulations purely foreign phenomena?
-
The accounts receivable clerk for Vandalay Industries prepared the following partially completed aging-of-receivables schedule as of the end of business on November 30: The following accounts were...
-
Write a code to test a Gaussian pseudorandom number generator. If you do not have a canned generator available, write a generator based on the Box-Muller algorithm in Appendix I. Apply the following...
-
Genuine Spice Inc. began operations on January 1, 2014. The company produces a hand and body lotion in an eight-ounce bottle called Eternal Beauty. The lotion is sold whole-sale in 12-bottle cases...
-
Pops Popcorn has three project choices for the coming year, but only $9,000 in its budget for new projects. Project 1 is a new corn seed separator that identifies grannies (seeds that do not pop when...
-
By the method of combining reduction half-cell reactions illustrated, determine E l rO2 / Ir , given that E lr3+ / Ir = 1.156 V and E IrO2 / Ir 3+ = 0.223 V.
-
The theoretical voltage of the aluminumair battery is E cell = 2.71 V. Use data from Appendix D and equation (19.28) to determine f G for Al[(OH) 4 ] - . Eq. 19.28 4 Al(s) + 3 O(g) + 6 HO(1) + 4...
-
What do you think of these rules? Do you agree or disagree? Why?
-
A decision maker is faced with three alternatives, A, B, and C. The utility for alternative A is greater than the utility for alternative B, which is greater than the utility for alternative C. This...
-
The IS/LM/BP model captures the open economy explanation of the real effects of economic policy through changes in the exchange rate. 1). Assuming perfect capital mobility, discuss the rationale for...
-
A critical review combining available literature on the 'Dyson Electric Car Project' and current academic research literature to consider the following points: 1. The impact of leadership style and...
-
Read the case study " Walmart, Inc. in 2018: The World's Biggest Retailer Faces New Challenges" How successful is Walmart? How much of its success can be attributed to industry allure and how much to...
-
Janis is a retail sales associate at Canada's Home Store. She has not been meeting her sales targets and recently got into an argument with a customer over returns of products which were eligible for...
-
Star Corporation issued both common and preferred stock during 20X6. The stockholders' equity sections of the company's balance sheets at the end of 20X6 and 20X5 follow. a. Compute the number of...
-
With your classmates, form small teams of skunkworks. Your task is to identify an innovation that you think would benefit your school, college, or university, and to outline an action plan for...
-
The cash flows from operating activities are reported by the direct method on the statement of cash flows. Determine the following: (a) If sales for the current year were $685,000 and accounts...
-
The cost of merchandise sold for Kohl's Corporation for a recent year was $9,891 million. The balance sheet showed the following current account balances (in millions): Determine the amount of cash...
-
Selected data taken from the accounting records of Lachgar Inc. for the current year ended December 31 are as follows: During the current year, the cost of merchandise sold was $448,500, and the...
-
According to this case study, which of these product lines (s)( clothes, toys, or books) need sales promotions? Why does this product line or lines need sales promotion? DMC Case Study DMC is a...
-
Because your demand has an upward trend, you want to use a forecast including a trend. In period 18, the level is estimated at F18 = 5,520, and the trend is estimated at T18 = 40. These give you FIT...
-
Assume that every time you buy an item of the Hong Kong Disney series, you receive one of the four types of cards, each with a cartoon character Mickey, Minnie, Donald and Daisy with an equal...

Study smarter with the SolutionInn App


