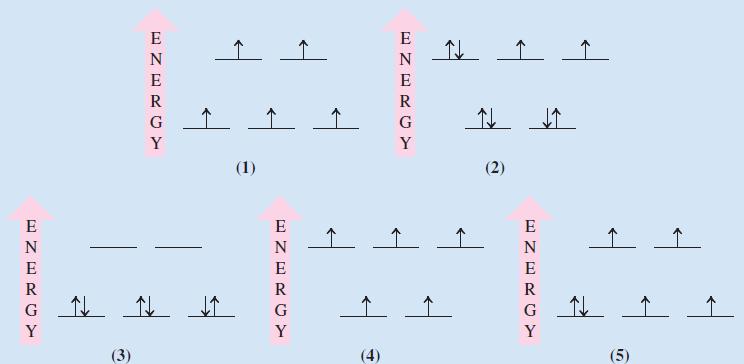
From the following crystal field splitting diagrams identify: (i) A tetrahedral Mn 2+ complex; (ii) A strong-field
Question:
From the following crystal field splitting diagrams identify:
(i) A tetrahedral Mn2+ complex;
(ii) A strong-field octahedral complex of Co3+;
(iii) A weak-field octahedral complex of Fe2+;
(iv) A tetrahedral Ni2+ complex;
(v) A high-spin octahedral complex of Fe3+.
Transcribed Image Text:
E N E R G Y N (3) EN E R G Y N (1) EN RGY ↑ (4) E R G Y N (2) E N E R G Y (5)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Based on the crystal field splitting diagrams providedthe following can be identified Crystal field ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The [Ni(CN)4]2- ion, which has a square-planar geometry, is diamagnetic, whereas the [NiCl4]2- ion, which has a tetrahedral geometry, is paramagnetic. Show the crystal field splitting diagrams for...
-
Cebo Zikode started Formula Trucks Inc in 1999 and is famous in the racing business and has worked with many different suppliers. Formula Trucks Inc. manufactures high quality trucks, purchases and...
-
For the following crystal field diagrams, label each as low spin, high spin, or cannot tell. Explain your answers. a. b. c.
-
If the molecular weight of air is 28.9, what is the density of air at atmospheric pressure and a temperature of 328.2 K? 1 atm = 1.013 x 10 5 N/m, the mass of a proton is 1.67262 x 10 -27 kg,...
-
Big Company purchased Little Company for $290,000. At the time of purchase, the fair value of Little Companys assets and liabilities was as follows: Inventory . . . . . . . . . . . . . . . . . . . ....
-
List the five primary activities involved in the acquisition and payment cycle.
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the ERR for this project. b. Is this project economically attractive? EOY 0 2 3 4 5 6 NCF -$100 $15 $15 $15...
-
Victoria Companys balance sheet at December 31, 2013, is presented below. During January 2014, the following transactions occurred. Victoria uses the perpetual inventory method. Jan. 1 Victoria...
-
Explain an existing healthcare initiative that address the disparities veterans face with access to mental health. Explain specific goals of the specific initiative to address the gap veterans face...
-
Draw a plausible structure to represent: (a) [FeBr(ox) 2 ] 2 (b) [CoBr 4 (NH 3 ) 2 ] (c) [Cu(EDTA)] 2 (d) [CrBr 2 (H 2 O) 4 ] + (e) [PtBr 6 ] 2 .
-
(A) The color of [Co(H 2 O) 6 ] 2+ is pink, whereas that of tetrahedral [CoCl 4 ] 2 is blue. Explain this difference in color. (B) One of the following solids is yellow, and the other is green: Fe(NO...
-
Assuming negligible damping (j = 0), calculate the group velocity (vg = dw/dk) of the waves described by Eqs. 9.166 and 9.169. Show that vg < c, even when v > c.
-
Many of the buildings in Paris have Mansard roofs, such as those shown in the photograph on page 147. a. What property tax structure would bring this about? b. Could you imagine a change in the...
-
In the 1960s, coffee came in 1-pound cans. Today, most coffee comes in 11-ounce cans. a. Can you think of an explanation why? b. Can you think of other products besides coffee whose standard size has...
-
Why would an economist be more hesitant about making an elasticity estimate of the effect of an increase in price of 1 percent than an increase in price of 50 percent?
-
Which of the following producers would you expect to support a tax on beer? Which would not? Explain your answer. a. Producers of hard liquor. Cross-price elasticity with beer: 0.11. b. Producers of...
-
Suppose average movie ticket prices are $8.50 and attendance is 1.2 billion. The price of tickets rises to $9.50 and attendance rises to 1.4 billion. a. What happened to total revenue? b. If you were...
-
What is the basis for valuing assets transferred by gift and at death?
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
The unadjusted trial balance for Sierra Corp. is shown in Illustration 4-4 (page 168). In lieu of the adjusting entries shown in the text at October 31, assume the following adjustment data. 1....
-
The income statement of Kaleta Co. for the month of July shows net income of $1,500 based on Service Revenue $5,500; Salaries and Wages Expense $2,100; Supplies Expense $900, and Utilities Expense...
-
This is a partial adjusted trial balance of Fenske Company. InstructionsAnswer these questions, assuming the year begins January 1.(a) If the amount in Supplies Expense is the January 31 adjusting...
-
3.) The Buckingham Pi Theorem allows one to create dimensionless functions and groups to make data more universally useful. This problem begins with a dimensional function, named f. Its arguments and...
-
1. (30%) Consider a dynamic system described with the following model. 100 [0] z= 0 a bz+Ou 1 22 (a) Find values a, and b that make the system stable with eigenvalues -1, -1 and -2. (b) Find the...
-
Central banks' policies during the global financial crisis in 2008 were designed to: Question 4Answer a. Increase liquidity in the market b. Start purchasing bonds from the private sector c. Act as...

Study smarter with the SolutionInn App


