How many elements of unsaturation are there in the molecule below? What is the molecular formula? HN
Question:
How many elements of unsaturation are there in the molecule below? What is the molecular formula?
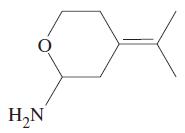
Transcribed Image Text:
H₂N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate the elements of unsaturation also known as degrees of unsaturation or double bond equiv...View the full answer

Answered By

Fozia tabasum
I am in Doctorate, Civil Engineering
0.00
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
How many elements of unsaturation are there in the molecule below? What is the molecular formula? Br CN
-
Consider the following molecular formulas. How many elements of unsaturation are there in each case? (a) C 5 H 9 NO (b) C 5 H 8 O 3 ; (c) C 5 H 9 ClO.
-
Consider the following molecular formulas. How many elements of unsaturation are there in each case? (a) C 4 H 11 N; (b) C 4 H 6 O; (c) C 9 H 15 ClO.
-
Find the values of the parameter p > 0 for which the following series converge. 00 1 k=2 (Ink)P
-
Which one of the following statements is incorrect? a. Sunk costs should be irrelevant in decision making. b. Differential costs are the costs a company should consider when making decisions. c....
-
It has been determined that, in aggregate, financial institutions with depository accounts currently hold excess reserves equal to $3 billionthat is, they hold $3 billion more than is necessary to...
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
Summerborn Manufacturing, Co. completed the following transactions during 2014: Jan. 16 Declared a cash dividend on the 5%, $ 100 par noncumulative preferred stock (900 shares outstanding). Declared...
-
How did you apply active listening to seek information and confirm your understating as you interacted with a child or group of children?
-
Match the following compounds with the chemical properties in the next column. Write a chemical equation for the reactions described in (a)-(d). (a) Is easily oxidized; (b) Decolorizes bromine water;...
-
Draw the structure for each of the following. (a) (R)-1-bromo-1-chloroethane (b) (E)-2-bromopent-2-ene (c) (Z))-1-chloro-3-ethylhept-3-ene (d) (R)-2-hydroxypropanoic acid (e) (S)-2-aminopropanoate...
-
Let S = $40, K = $40, r = 8% (continuously compounded), = 30%, = 0, T = 0.5 year, and n = 2. a. Construct the binomial tree for the stock. What are u and d? b. Show that the call price is $4.110....
-
Discuss the components of consolidated financial statements. Describe the purpose of consolidated financial statements by giving 2 examples. Discuss the component that provides information to analyze...
-
What is the term for the value of goods and services and the financial assets and liabilities flowing between the U . S . and other countries?
-
Which one of the following operating lease disclosures are required to be presented by a lessor under IFRS 1 6 ? Separate disclosure of income relating to variable lease payments that do not depend...
-
During the year, the Senbet Discount Tire Company had gross sales of $895,000 million. The firms cost of goods sold and selling expenses were $461,000 and $215,000, respectively. Senbet also had...
-
The labor-force participation rate equals the percentage of the total adult population that is in the labor force a. True b. False
-
Ms. M was born in 1918. After retiring, Ms. M returned to Fairfield, South Carolina, where she lived on her family farm with her brother, a dentist, until his death in the early 1980s. Ms. M never...
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
What is Computrons free cash flow (FCF)? What are Computrons net uses of its FCF?
-
Calculate Computrons return on invested capital. Computron has a 10% cost of capital (WACC). Do you think Computrons growth added value?
-
Jamison also has asked you to estimate Computron's EVA. She estimates that the after-tax cost of capital was 10 percent in both years.
-
Describe a criminal offense from the California Penal Code and the corresponding CALCRIM jury instruction setting out the elements of that offense. Identify whether that offense is a specific or...
-
Harold is relieved - the Unit Chair did not notice that he had submitted Martha's work as his own assessment task in his History of Law unit and he ultimately received a good mark for it. A few years...
-
Alice is a solicitor in Victoria. Alice runs her own law firm which specialises in criminal law. She often appears in the Victorian Magistrates' Court on behalf of her clients. There is a particular...

Study smarter with the SolutionInn App


